PNAS: Ferroptosis is a powerful way to treat cancer
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
PNAS: Ferroptosis is a powerful way to treat cancer
PNAS: Ferroptosis is a powerful way to treat cancer. Ferroptosis is a necrotic cell death catalyzed by iron ions. It is characterized in that when the intracellular reduction system is inactivated, the phospholipid molecules containing the long chain of unsaturated fatty acids on the cell membrane or organelle membrane are destroyed by peroxide, causing the cell membrane to rupture. This form of cell death was named Ferroptosis in 2012.
Ferroptosis is tightly regulated by intracellular signaling pathways. Ferroptosis is mainly regulated by glutathione peroxidase 4 (GPX4). Once GXP4 is abnormal, it will lead to decreased cellular antioxidant capacity, accumulation of reactive oxygen species, and ultimately cell Ferroptosis.
Ferroptosis is related to Parkinson’s disease, pancreatic cancer, diabetic complications, renal failure, cardiovascular and cerebrovascular diseases and other diseases. It has been discovered that Ferroptosis can be activated or inhibited to interfere with the development of the disease. Therefore, Ferroptosis has become a recent phenomenon. Research hotspots.
As we all know, cancer cells grow vigorously and have a stronger ability to divide and reproduce, so they have a stronger demand for metabolism, but this feature also provides clues for their elimination. Gene mutations that enable cancer cells to cope with oxidative stress make them more resistant to Ferroptosis. Without these mutations, cancer cells may be very sensitive to Ferroptosis.
Cancer cells multiply fast, which greatly increases the demand for metabolism. The PI3K-AKT-mTOR pathway regulates metabolism, and this pathway is also one of the most common mutations in cancer. At the same time, cancers with mutations in the PI3K-AKT-mTOR pathway are also One of the most difficult types of cancer to treat.
Recently, the team of Professor Xuejun Jiang from the Memorial Sloan Kettering Cancer Center in the United States published a titled “Oncogenic activation of PI3K-AKT-mTOR signaling suppresses ferroptosis via SREBP-mediated lipogenesis” in the Proceedings of the National Academy of Sciences (PNAS).
Research papers.
The research team found that the metabolic changes often exhibited by cancer cells make them vulnerable to
The research team found that the combination of drugs that block the PI3K-AKT-mTOR pathway and drugs that induce Ferroptosis can significantly destroy and eliminate tumors due to the effects of ferroptosis, a special type of cell death.

The research team found that in in vitro cell experiments, tumor cells with mutations in the PI3K-AKT-mTOR pathway showed strong resistance to drugs that induce cell Ferroptosis.
Next, the research team will use a combination of drugs that block the PI3K-AKT-mTOR pathway and drugs that induce cell Ferroptosis, and these cancer cells will die.
Then, the research team conducted verification in mouse models of breast and prostate cancer that contain mutations in the PI3K-AKT-mTOR pathway. Experimental results showed that the combination of these two drugs can almost completely destroy and eliminate tumors in mice.
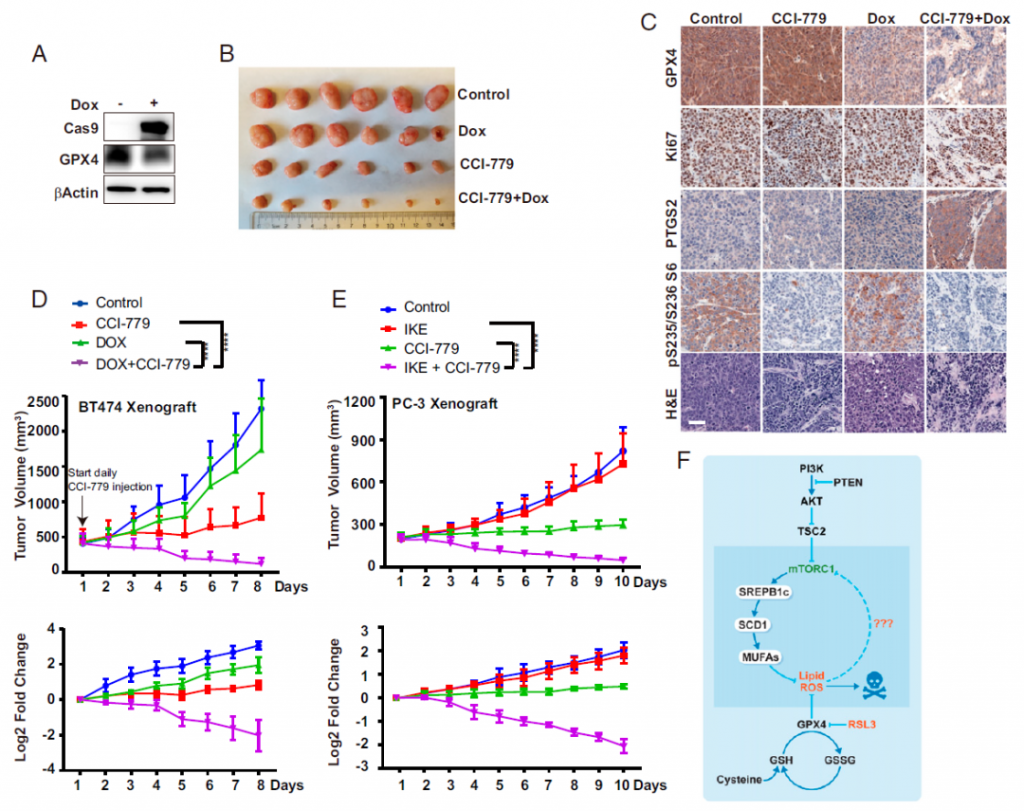
Faced with the results of this experiment, Professor Jiang Xuejun, the corresponding author of the study, said that this is the most significant tumor regression that has never been seen in his laboratory.
The research team further showed that the mutant PI3K-AKT-mTOR pathway can increase the activity of proteins involved in cell membrane lipid synthesis, thereby increasing cell membrane lipids and protecting cells from Ferroptosis. By blocking the PI3K-AKT-mTOR pathway, it is possible to pump down, prevent excessive synthesis of lipids, and make cells sensitive to Ferroptosis.
On July 24, 2019, Jiang Xuejun’s team and others published a paper in Nature and found that certain gene mutations in the Hippo pathway can make cells more sensitive to Ferroptosis providing a new research direction for cancer treatment through Ferroptosis.

This study by PNAS is a supplement and development to the Nature paper mentioned above. Gene mutations in many cancers are related to the sensitivity of cells to Ferroptosis. Therefore, these mutations can be used to initiate Ferroptosis, thereby killing cancer cells. It is expected to become a new and exciting cancer treatment method.

It is reported that Professor Xuejun Jiang’s team has applied for a patent related to the research, and the next step will be to test the drug combination on tumor samples from cancer patients treated at the Memorial Sloan Kettering Cancer Center.
(source:chinanet, reference only)
Disclaimer of medicaltrend.org



