PNAS: The mechanism of organ selective mRNA delivery system
- Aspirin: Study Finds Greater Benefits for These Colorectal Cancer Patients
- Cancer Can Occur Without Genetic Mutations?
- Statins Lower Blood Lipids: How Long is a Course?
- Warning: Smartwatch Blood Sugar Measurement Deemed Dangerous
- Mifepristone: A Safe and Effective Abortion Option Amidst Controversy
- Asbestos Detected in Buildings Damaged in Ukraine: Analyzed by Japanese Company
PNAS: The mechanism of organ selective mRNA delivery system
PNAS: The mechanism of organ selective mRNA delivery system, greatly expanding the application scope of mRNA and CRISPR technology.
In recent years, as a new pharmaceutical technology , mRNA has made breakthrough progress in the field of infectious diseases and tumor treatment in a short period of time.
However, how to safely and efficiently deliver mRNA drugs to specific target cells and protect them from degradation is one of the main obstacles to current mRNA therapy.
The ideal delivery vehicle must be safe, stable and organ-specific. Lipid nanoparticles (LNP) are currently the most advanced mRNA delivery vector in clinical practice.
At present, all new coronavirus mRNA vaccines that are being developed or approved for clinical use are delivered using LNP vectors.
LNP provides many benefits for mRNA delivery, including simple formulation, modularity, biocompatibility and larger mRNA payload.
However, clinical studies have shown that LNP accumulates in the liver , so most of the current LNP delivery systems are liver-targeted, and the problem of effective delivery to organs outside the liver (such as the lung and kidney) needs to be solved urgently.
December 28, 2021, American University of Texas Southwestern Medical Center Daniel Siegwart , Professor, Cheng Jiang Boshi (now the Peking University Institute of Technology researcher future) as co-corresponding author, in ” American Academy of Sciences ” (PNAS) published entitled: the On at the mechanism of Tissue-specific mRNA Delivery by Selective Targeting nanoparticles Organ research paper [1] .
The team previously developed a selective organ targeting lipid nanoparticle and named it SORT (selective organ targeting) . By adding a new lipid SORT lipid, it can be specific to organs such as liver, lung, spleen, etc. Sexual targeting .
This study further analyzed the specific mechanism by which this selective organ-targeting lipid nanoparticles can achieve tissue-specific delivery. This will greatly expand the application range of mRNA vaccines and drugs, as well as CRISPR gene editing therapies.

Lipid nanoparticles (LNP) usually include four components, ionizable lipids, cholesterol, auxiliary phospholipids, and pegylated lipids.
These components and mRNA form nanoparticles in acidic buffer to encapsulate and protect fragile mRNA. . In addition, they are positively charged in the acidic environment of endosomes, which promotes their fusion with endosome membranes and releases them into the cytoplasm.
Before the publication of this PNAS paper, Professor Daniel Siegwart ’s team found that adding the fifth component- SORT lipids to lipid nanoparticles can change the organ targeting properties of LNP in the body and achieve mRNA targeting to organs other than the liver. deliver.
More importantly, this SORT-LNPs can be extended to a variety of extrahepatic organs and tissues to achieve mRNA delivery to lungs, kidneys, and even epithelial cells and immune cells.
The findings in April 2020 published in Nature Nanobitechnology journal [2] , Qiang Cheng (now the Peking University Institute of Technology researcher future) , Wei properly (now a researcher at the Chinese Academy animal) is co-first author of the paper.

The papers have been published in the medical field has aroused widespread concern, but that the mechanism of action SORT-LNPs remains unclear.
Therefore, in this PNAS paper, in order to understand how SORT-L NPs can break through the delivery barrier accumulated in the liver, the research team investigated the mechanistic factors that define its organ targeting characteristics.
The research team first identified and studied the mechanism factors that can explain the organ-targeting properties of SORT-LNPs based on the three established principles for determining the efficacy of liver-targeted LNPs: organ-level biodistribution, acid dissociation constant (pKa), and serum protein adsorption .
The research team found that these three factors are different in SORT-LNPs targeted by different organs and are related to their tissue targeting properties.
For example, the acid dissociation constant (pKa) of liver-targeted LNPs is close to 6.4, and the adsorption of apolipoprotein E (ApoE) on the surface of LNPs .
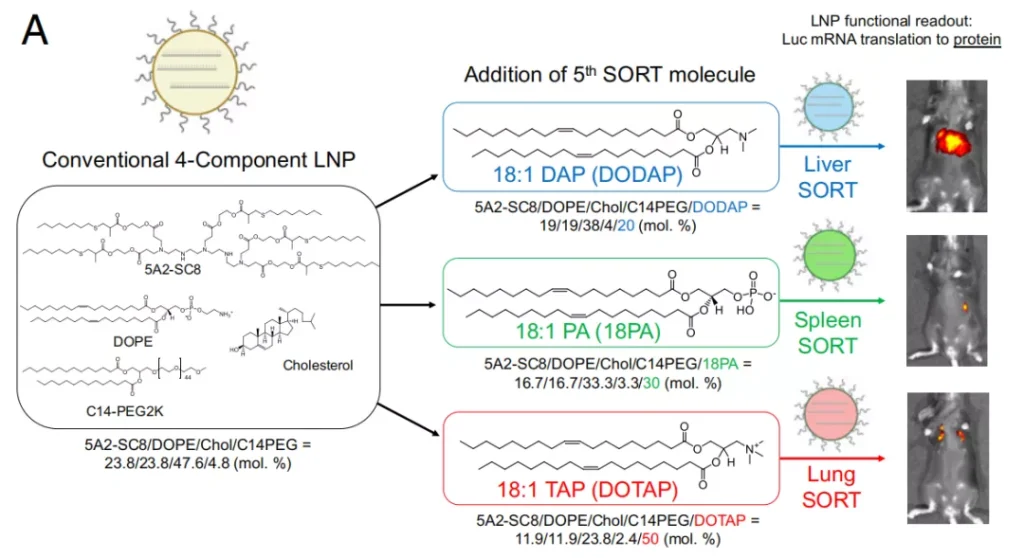
SORT nanoparticles for tissue-specific mRNA delivery have unique biodistribution and ionization behavior
In addition, the research team provided evidence of the three-step mechanism of the functional role of serum proteins in tissue targeting:
First, the desorption of PEG lipids on the surface of LNP exposed the potential SORT molecules in LNP.
Next, different serum proteins recognize the exposed SORT molecules and adsorb them on the surface of the LNP.
Finally, surface-adsorbed proteins interact with homologous receptors expressed by cells in target organs to promote the functional delivery of mRNA to these tissues.
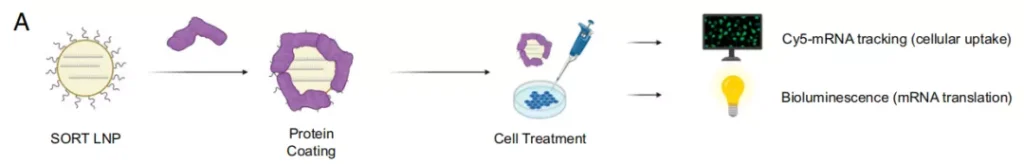 Different SORT molecules adsorb different serum proteins
Different SORT molecules adsorb different serum proteins
These findings establish a key connection between the molecular composition of SORT nanoparticles and their unique and precise organ targeting properties, and show that the choice of SORT molecules determines which proteins can be adsorbed on the surface of LNPs, thereby affecting SORT-LNPs The delivery destination.

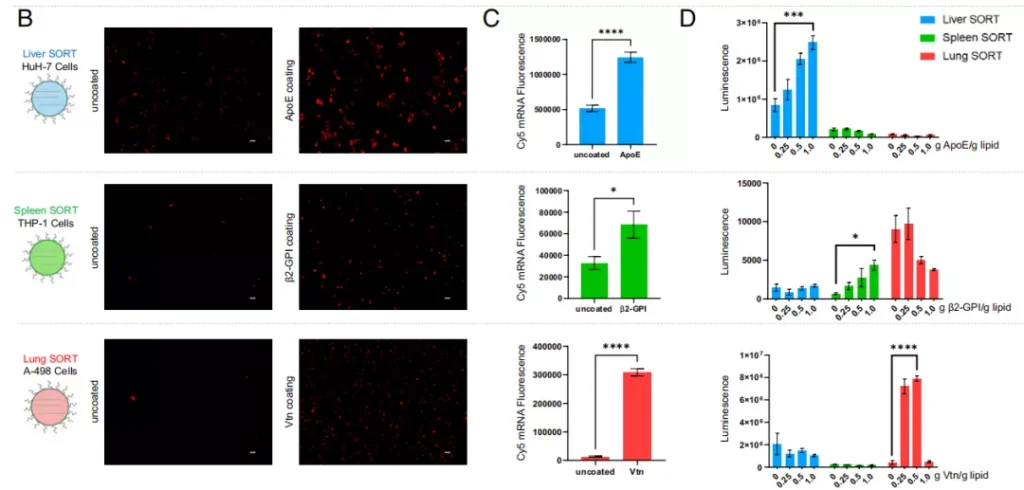
SORT molecules adsorb different serum proteins, thereby determining the focus of SORT-LNPs delivery
All in all, this study explains the specific mechanism of SORT-LNPs to achieve specific delivery of organs and tissues , which will promote the optimization of SORT-LNPs in the treatment of lung, liver, spleen and other tissues and organs, and will help expand the SORT platform to other nanoscale The type of particles, physiological tissues and cell types lay the foundation.
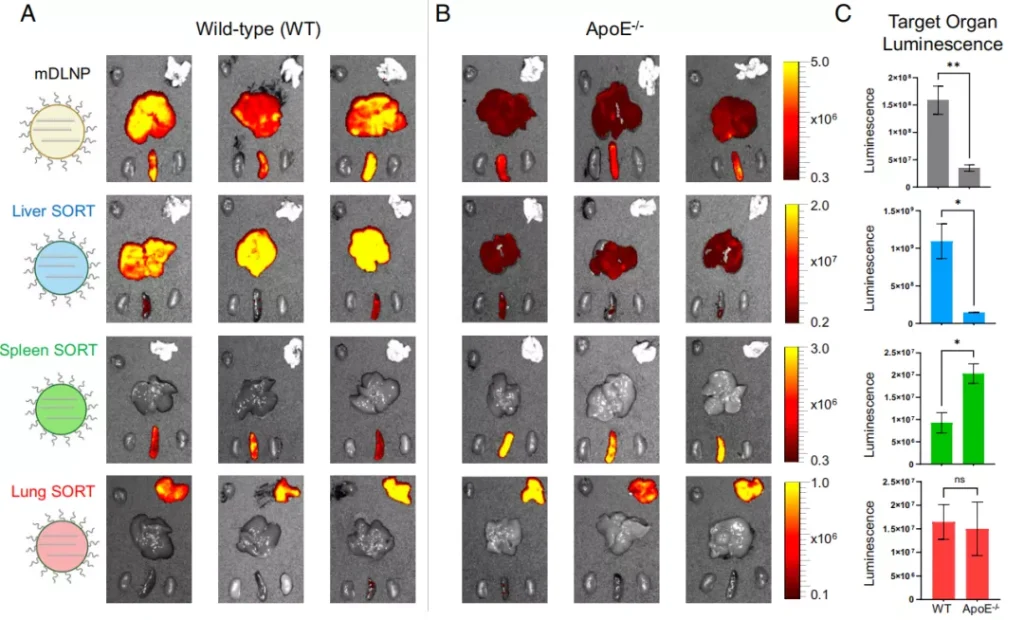 Extrahepatic mRNA delivery is carried out through a mechanism that does not depend on ApoE protein
Extrahepatic mRNA delivery is carried out through a mechanism that does not depend on ApoE protein
According to the author, the results of this study show that endogenous targeting-that is, adjusting the molecular composition of nanoparticles to bind to specific proteins in the serum so that they can be delivered to the target site.
This may be an effective strategy to achieve extrahepatic delivery of nanocarriers.
Reference:
1.https://www.pnas.org/content/118/52/e2109256118
2. https://www.nature.com/articles/s41565-020-0669-6
PNAS: The mechanism of organ selective mRNA delivery system
(source:internet, reference only)
Disclaimer of medicaltrend.org



