Can CAR-T Therapy Really Cure Cancer after one decade passed?
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Can CAR-T Therapy Really Cure Cancer after one decade passed?
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Can CAR-T Therapy Really Cure Cancer after one decade passed?
In recent years, CAR-T therapy has achieved encouraging results in various malignant tumors, especially hematologic malignancies, bringing new hope to patients. However, the clinical application of CAR-T therapy is complex, and its long-term efficacy and safety still need validation.
On May 10, 2023, Emily, a renowned cancer survivor and the world’s first recipient of CAR-T therapy, celebrated the 11th year of cancer-free survival.
Recently, Professor James N. Kochenderfer from the National Cancer Institute published a comprehensive review titled “Long-term outcomes following CAR T-cell therapy: what we know so far” in the prestigious journal Nature Reviews Clinical Oncology.
The review summarizes and analyzes the structural design of CAR-T products on the market for the past decade, the long-term therapeutic effects of CAR-T therapy in different hematologic tumors, factors influencing treatment response, and safety considerations.
CD19 CAR-T Therapy in Relapsed/Refractory B-cell Lymphoma and Chronic Lymphocytic Leukemia
Clinical trials involving 10 CD19 CAR-T products in patients with relapsed/refractory B-cell lymphoma and chronic lymphocytic leukemia, with follow-up data ranging from 24 to 123 months, showed an overall response rate of 44-91% and a complete response rate of 28-68%.
Notably, some patients maintained deep remission without further consolidation therapy for more than 2 years post-infusion.
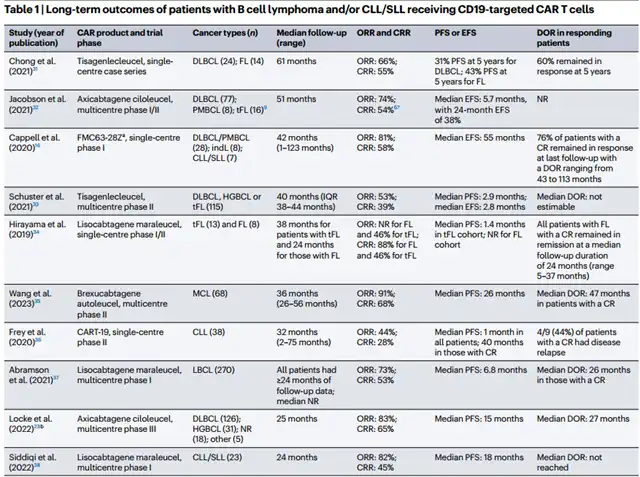
Recent follow-ups revealed sustained complete responses exceeding 43 months, with one trial using axicabtagene ciloleucel showing a complete response rate of 55%, with 60% of patients still in remission after 5 years.
These findings suggest the potential for CD19 CAR-T therapy to cure relapsed/refractory B-cell lymphoma and chronic lymphocytic leukemia without additional interventions.
CD19 CAR-T Therapy in B-cell Acute Lymphoblastic Leukemia (B-ALL)
Twelve clinical trials involving CD19 CAR-T in B-ALL patients showed promising initial complete response rates (62%-86%).
However, the duration of event-free survival varied among studies, considering the heterogeneity of included patients.
The study emphasizes that the significance of allogeneic hematopoietic stem cell transplantation (allo-HSCT) after CAR-T therapy differs between pediatric and adult B-ALL patients.
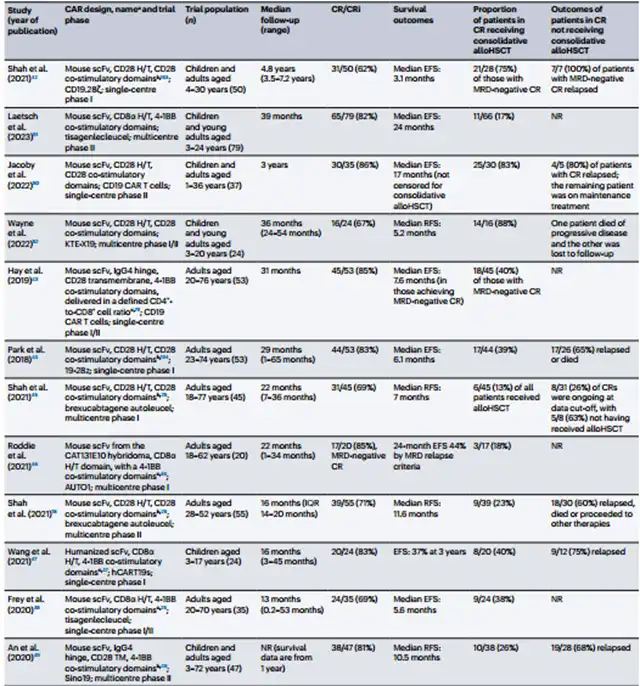
Overall, CD19 CAR-T therapy in B-ALL patients demonstrated high complete response rates, but pediatric patients showed suboptimal sustained event-free survival compared to B-cell lymphoma patients, with a lower proportion achieving cure without further consolidation therapy.
BCMA CAR-T Therapy in Relapsed/Refractory Multiple Myeloma (R/R MM)
Compared to CD19 CAR-T, BCMA CAR-T research started later. Six clinical studies with long-term follow-up (range: 13-48 months) in R/R MM patients revealed impressive overall response rates (73-100%) and complete/sustained complete response rates (33-83%).
Some patients maintained prolonged progression-free survival without further consolidation/maintenance therapy for over a year.
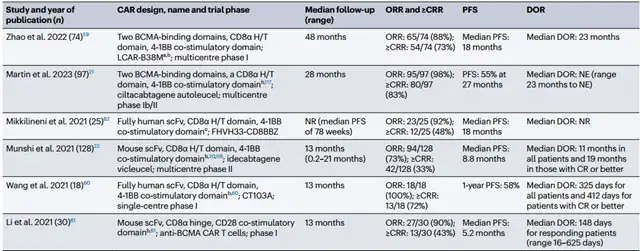
However, a study using fully human BCMA CAR-T with only heavy chain antigen recognition reported a median progression-free survival of 18 months without maintenance therapy. The study highlights the ongoing risk of disease progression over time.
The research concludes with six factors influencing CAR-T therapy efficacy:
- Depth of Remission: Patients with a good initial response are more likely to achieve long-term remission.
- Disease Type: B-cell lymphoma patients exhibit a lower complete response rate, but once achieved, remission is more likely to be sustained. B-ALL patients have a higher complete response rate but a lower sustained remission rate.
- Tumor Burden and Extramedullary Disease: Patients with smaller tumor burdens/volumes pre-infusion are more likely to achieve deep remission. Patients with extramedullary disease have a poorer treatment response.
- Pre-infusion Lymphodepletion: Patients receiving lymphodepletion before treatment may achieve deeper remission.
- Post-infusion CAR-T Expansion Levels: Higher levels of blood CAR-T expansion correlate with better initial response and deep remission, indicating long-term presence in the body.
- Post-infusion MRD Status: Even MRD-negative patients may experience disease relapse.
In conclusion, the review provides valuable insights into the progress and challenges of CAR-T therapy over the past decade, emphasizing the need for continued research and optimization of treatment strategies for different cancer types and patient populations.
Can CAR-T Therapy Really Cure Cancer after one decade passed?
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



