BRCA1/2 gene detection can prevent millions of breast and ovarian cancers!
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
BRCA1/2 gene detection can prevent millions of breast and ovarian cancers!
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
“Cancers”: BRCA1/2 gene detection can prevent millions of breast and ovarian cancers!
Breast and ovarian cancers are the most common cancers in women.
BRCA1 and BRCA2 are the “star genes” that cause breast cancer and ovarian cancer . About 6% of breast cancer and 10-20% of ovarian cancer are caused by heritable BRCA1/2 gene mutations.
Before the age of 80, women with BRCA1/2 mutations have a 69-72% risk of developing breast cancer and a 17-44% risk of developing ovarian cancer.
If mutation carriers can be screened out through BRCA1/2 gene testing before they get sick, millions of cancers (breast/ovary cancer) could be prevented with drugs, increased screening, or surgery.
Currently, global clinical guidelines only recommend BRCA1/2 gene testing for high-risk women (age of diagnosis and family history of certain cancers, etc.). Carriers of BRCA1/2 germline mutations.
On July 17, 2020, an international study published in “Cancers” showed that, compared with current clinical practice, screening the entire population for breast and ovarian cancer susceptibility genes BRCA1/2 mutations can prevent global Breast and ovarian cancers occur within the millions.

The researchers simulated a population-based BRCA1/2 detection model (Markov model), analyzed the data of ordinary women aged ≥ 30 years in six countries with different incomes in the United States, the United Kingdom, the Netherlands, China, Brazil, and India, and analyzed Life cycle costs and health impacts were compared with current clinical criteria/family history based testing.
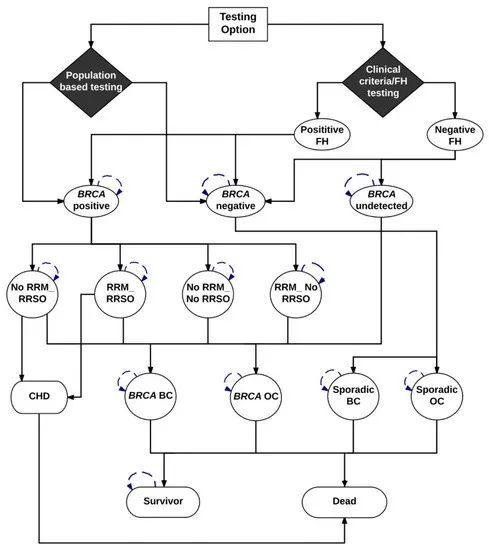 ▲ Markov model
▲ Markov model
The World Bank uses GNI per capita (USD) to classify countries into four income categories: low-income (LIC ≤ 1,025$); lower-middle-income (LMIC: 2,026$-4,035$, such as India); -12,475$, such as China/Brazil); high income (HIC: ≥12,476$, such as US/UK and Netherlands)
Cost-effectiveness is calculated from a social and payer perspective. The payer perspective includes only medical costs borne by the health system or health providers (eg, costs of genetic testing, screening, prevention, and cancer treatment).
From a societal perspective, costs are also taken into account, such as the impact of lost income due to inability to work and shortened lifespan due to cancer.
Findings suggest that BRCA1/2 genetic testing for all women aged ≥30 years and classification as BRCA1/2 positive or BRCA1/2 negative is cost-effective and highly cost-effective from a payer perspective in upper-middle-income countries Cost-effectiveness.
In the United Kingdom, the United States and the Netherlands, the incremental cost-effectiveness ratio (ICER, i.e., each additional QALY The extra costs required) are: USD 21,191/QALY, USD 16,552/QALY and USD 25,215/QALY; the incremental cost-benefit ratios in China and Brazil are: USD 23,485/QALY and USD 20,995/QALY respectively.
It is not cost-effective to conduct general screening of BRCA1/2 in India, and the incremental cost-benefit ratio is US$32,217/QALY. In low- and middle-income countries like India, the cost of BRCA1/2 testing needs to come down to around $172 to be cost-effective.
The evaluation of medical economics uses the incremental cost-benefit ratio (ICER) to characterize the evaluation results, namely: cost difference/effect difference, and the effect usually uses quality-adjusted life years (QALYs) as an indicator. After obtaining ICER, the economical efficiency of health technology is judged by comparing with the cut-off value.
The threshold value is US$50,000 in developed countries in Europe and the United States, and 3 times (or lower) per capita GDP in developing countries, which reflects the cost that society is willing to pay for an additional quality-adjusted life year.
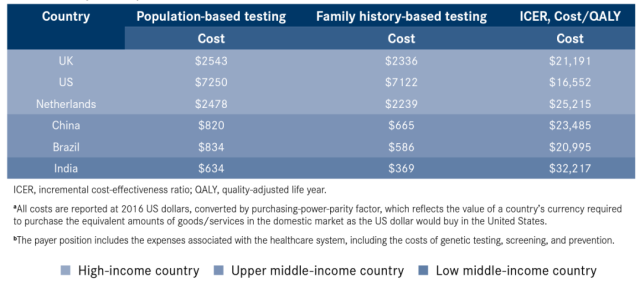
Cost of population-based and family history testing: cost savings from a payer perspective
The results of the study show that compared with current clinical practice, BRCA1/2 detection based on age ≥30 years old will prevent an additional 2,319-2,666 cases of breast cancer and 327-449 cases of ovarian cancer per million women.
- 57,708 cases of breast cancer and 9,700 cases of ovarian cancer could have been prevented in the UK;
- 269,089 cases of breast cancer and 43,817 cases of ovarian cancer could be prevented in the United States;
- In the Netherlands, 15,181 cases of breast cancer and 2,557 cases of ovarian cancer could be prevented;
- In China, 1,050,314 cases of breast cancer and 154,756 cases of ovarian cancer could be prevented;
- In Brazil, 156,299 cases of breast cancer and 25,170 cases of ovarian cancer could be prevented;
- 692,571 cases of breast cancer and 97,659 cases of ovarian cancer could be prevented in India;
In this way, the equivalent of preventing millions of breast and ovarian cancers in the entire population.
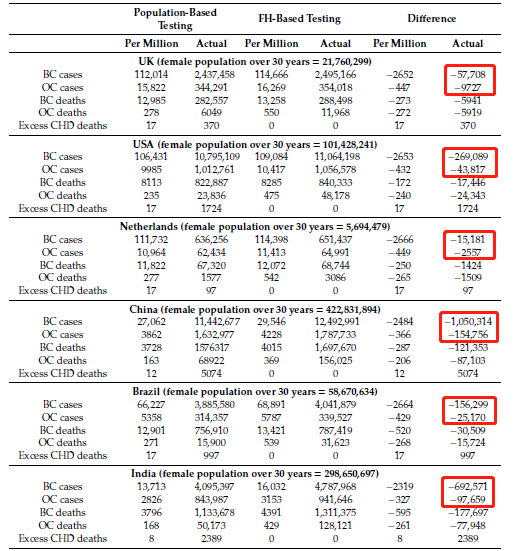
Most cancers in unaffected female carriers of BRCA1/2 mutations are preventable, the researchers believe .
For example, for ovarian cancer, you can choose risk-reducing salpingo-oophorectomy (RRSO), which can reduce the risk of ovarian cancer by 79%-96%;
For breast cancer, options include enhanced screening with MRI/mammography, prophylaxis with an estrogen receptor modulator (SERM) or aromatase inhibitor, or risk-reducing mastectomy (RRM) , can reduce the risk of breast cancer by 90-95%.
With the improvement of social awareness and acceptance of genetic testing, the decline of testing costs, and the advancement of computing technology and testing technology, it is possible to implement large-scale population testing.
This paradigm shift in population-based BRCA1/2 testing could ensure that more women take preventive measures to reduce their cancer (breast and ovarian) risk. Testing all women for BRCA1/2 mutations is a more cost-effective strategy.
References:
1. Manchanda Ranjit, Sun Li, Patel Shreeya et al. BRCA1/BRCA2 Economic Evaluation of Population-Based Mutation Testing across Multiple Countries and Health Systems. [J]. Cancers (Basel), 2020, 12: undefined.
BRCA1/2 gene detection can prevent millions of breast and ovarian cancers!
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



