The best weight loss drug in history?
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
The best weight loss drug in history?
The best weight loss drug in history? The phase-3 clinical trails success, once a week, 20% weight loss!
In today’s society, with economic development and improvement of living standards, obesity has become a major public health problem worldwide. According to statistics from the World Health Organization (WHO), nearly 2 billion people are overweight or obese worldwide. From 1975 to 2016, the global obesity rate has nearly tripled. Every year, overweight or obesity causes 2.8 million deaths.
In fact, obesity is not only a feature, but also a disease. Obese people not only have inconvenience in life and reduced exercise capacity, but are also more susceptible to metabolic diseases and cardiovascular and cerebrovascular diseases. In addition, many studies have shown that obesity is associated with more than ten kinds of cancers. The increased risk of disease is related to the decreased prognosis and survival rate.
On February 10, 2021, the top international medical journal “New England Journal of Medicine” (NEJM) published a paper that brought great news for those who need to lose weight.
The research team of University College London conducted a global large-scale clinical trial involving 1961 people in 16 countries around the world. The results of the trial showed that the new weight-loss drug semaglutide (semaglutide) was effective, and the participants in the clinical trial lost an average weight 15.3 kg.
Specifically, three-quarters of people lost more than 10% of their weight, and more than one-third of people lost more than 20% of their weight. This weight loss effect can be called a “game-changing”. The weight loss effect can only be achieved by surgery.
More importantly, people who lose weight after taking the new weight loss drug semaglutide (semaglutide) have reduced risk factors for heart disease and diabetes such as waist circumference, blood lipids, blood sugar, and blood pressure, and their overall quality of life has also been improved.

Human glucagon-like peptide 1 (GLP-1) is a brain-gut peptide secreted by human ileal endocrine cells. It is released into the blood from the intestine after a meal and can reduce hunger and increase satiety.
Semaglutide (Semaglutide), developed by Novo Nordisk, is a structural analogue of human glucagon-like peptide 1 (GLP-1) that can mimic its effects and has been approved for the treatment of type 2 diabetes.
Because semaglutide (Semaglutide) can reduce hunger, reduce diet, and reduce calorie intake, it is considered to be used to treat obesity.
The clinical trial called Semaglutide in the treatment of obese patients (STEP) has passed phase 1 and phase 2 clinical trials, but the safety of the 2.4 mg dose still needs to be evaluated.
This phase III randomized controlled clinical trial involves 1961 overweight or obese adults (none of them suffering from diabetes, with an average weight of 105 kg and an average body mass index of 38 kg/m2). The clinical trial was conducted in Asia, Europe, North America and South America Conducted at 129 locations in 16 countries.
Participants received a subcutaneous injection of 2.4 mg of semaglutide (or placebo) per week, which is similar to the way diabetes patients inject insulin. Overall, 94.3% of the participants completed the 68-week study beginning in the fall of 2018. Participants received face-to-face or telephone consultation and guidance from a registered dietitian every four weeks.
Patients treated with semaglutide (semaglutide) lost an average weight of 15.3 kg, and their average body mass index (BMI) dropped by 5.54. In the placebo group, the average weight loss was 2.6 kg, and the average BMI decreased by 0.92.
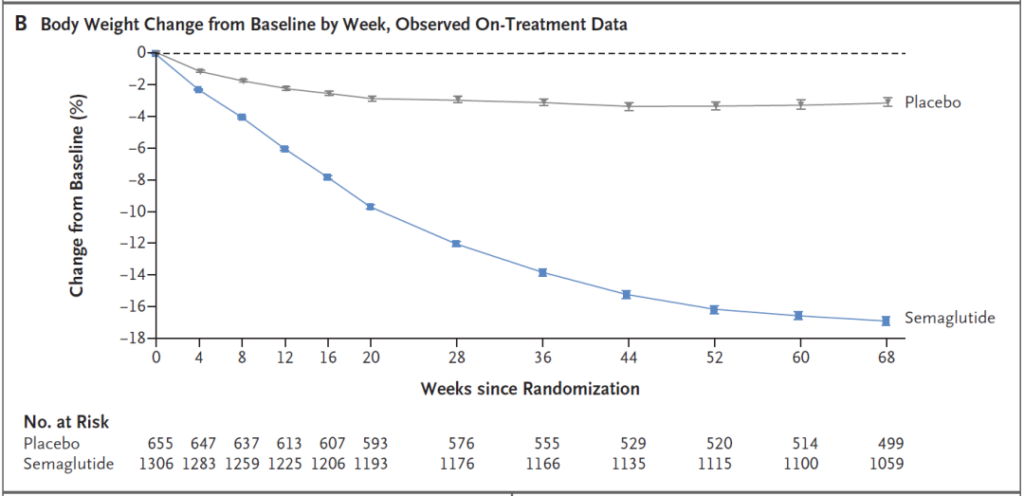
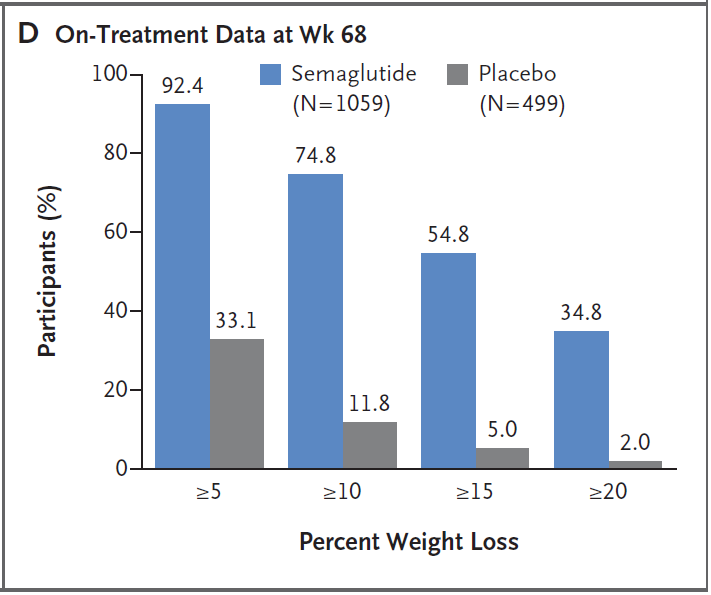
More importantly, people who lose weight after taking semaglutide have reduced risk factors for heart disease and diabetes such as waist circumference, blood lipids, blood sugar, and blood pressure, and their overall quality of life has also been improved.
In this phase 3 clinical trial, some participants reported side effects, including mild to moderate nausea and diarrhea, but these side effects are short-lived and can usually be resolved without the need to terminate the trial permanently.
This clinical trial represents a major advance in the treatment of obesity, because semaglutide (semaglutide) has been approved to treat type 2 diabetes in low doses, so doctors are already familiar with its usage.
With the results of this clinical trial as evidence, semaglutide (Semaglutide) has been submitted to the National Institute of Health and Clinical Optimization (NICE), the European Medicines Agency (EMA) and the US Food and Drugs as an obesity treatment drug The Supervision Administration (FDA) conducts regulatory approval.
(source:internet, reference only)
Disclaimer of medicaltrend.org



