Alzheimer’s disease: Toxic proteins continue to replicate
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Alzheimer’s disease: Toxic proteins continue to replicate
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Alzheimer’s disease: Toxic proteins continue to replicate.
Science Sub-Journal: Subversion of the understanding of the development process of Alzheimer’s disease, toxic proteins continue to replicate
In Alzheimer’s disease, tau protein and beta amyloid (Aβ) form tangles and plaques, which lead to brain cell death and brain atrophy, which can lead to memory loss, personality changes, and inability to take care of themselves. Approximately 44 million people worldwide are affected by Alzheimer’s disease, and as the global ageing intensifies, Alzheimer’s disease will also increase.
For many years, people have used terms such as “cascade” and “chain reaction” to describe the processes that lead to Alzheimer’s disease in the brain. Alzheimer’s disease is a disease that is difficult to study. The final diagnosis can only be made after examining the brain tissue samples after death, and animal models cannot reproduce tau protein and beta amyloid (Aβ) well. The rate of aggregation, which makes research progress very slow.
Recently, a research team from Cambridge University and Harvard University published a research paper titled: In vivorate-determining steps of tau seed accumulation in Alzheimer’s disease in Science Advances.
The study used human data for the first time to quantify the rate of different processes that cause Alzheimer’s disease, and found that its development method is completely different from previously thought. The toxin protein that was thought to cause Alzheimer’s disease forms aggregates in one area. , And then spread in the brain.
But this latest study shows that the toxin protein that causes Alzheimer’s disease does not start from a single point in the brain and triggers a chain reaction that causes brain cell death, but exists in different areas of the brain in the early stage, and over time Continue to replicate and expand over time. Therefore, trying to prevent the spread of toxic proteins between regions is of little help in slowing Alzheimer’s disease. The research helps develop new treatments for Alzheimer’s disease by targeting the most important processes that occur when humans develop.

The research team used brain samples from Alzheimer’s patients after death and PET scans from living patients (from patients with mild cognitive impairment to patients with full Alzheimer’s disease) to track the aggregates of tau protein. Based on the location and abundance patterns of tau aggregates observed in the brains of Alzheimer’s patients after death, they divided the disease into different Braak stages.
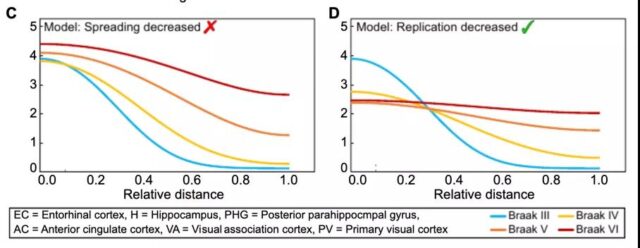
By combining five different data sets and applying them to the same mathematical model, the research team observed that most of the tau proteins in the later period of Braak are located in the entorhinal cortex, hippocampus and posterior hippocampal gyrus. In the early stages, tau appears to be limited to the entorhinal cortex and hippocampus, while the concentration in all other areas is relatively low.
After Braak 3, the distribution of tau protein changes more rapidly over time. It is worth noting that beta amyloid deposits in the neocortex and the appearance of disease symptoms often only occur in the later stage of Braak. Therefore, the research team focused on the stage where the disease progresses faster, which is Braak stage 3.
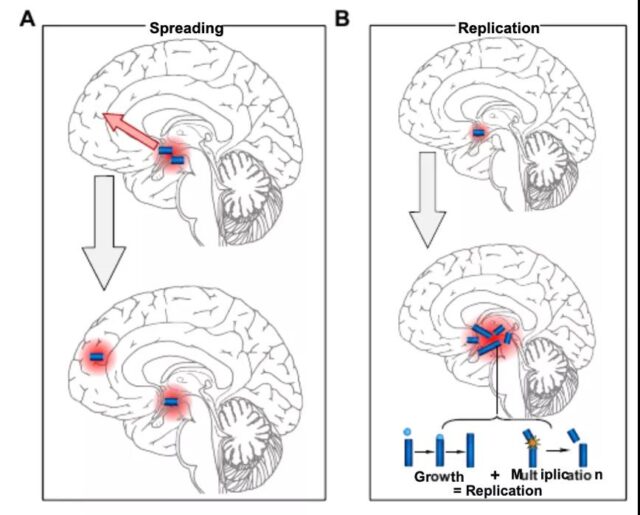
The research team discovered the importance of replication for the accumulation of tau protein. The reduction in replication significantly slowed the overall accumulation of tau. Therefore, the accumulation of tau protein is restricted by replication.
This means that after Braak 3, the overall rate of tau protein accumulation is determined by the rate of replication, and inhibition of replication will slow down the entire process to the greatest extent.
In order to quantify the aggregated tau protein, the research team used a variety of analysis methods: automatic image analysis of immunohistochemical slices; enzyme-linked immunosorbent assay; stereology technology; longitudinal tau-PET, which identified tau in Alzheimer’s disease The doubling time of protein is about 5 years, and it is expected to increase by about 100 to 1000 times within a few decades of the disease. This means that in Braak 3, there are already many aggregates widely distributed in the brain.
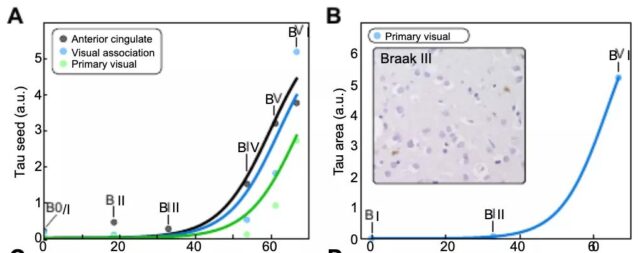
In summary, by applying chemical kinetics to human data, the study found that starting from Braak Phase 3, local replication, rather than spreading between brain regions, is the main factor controlling the total accumulation rate of tau protein in the neocortex.
The amount of tau protein only doubles every 5 years. Therefore, limiting local replication may be the most promising strategy to control the accumulation of tau protein during Alzheimer’s disease.
Original link:
https://www.science.org/doi/10.1126/sciadv.abh1448
Alzheimer’s disease: Toxic proteins continue to replicate
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



