Mahidol University develops chimeric Spike circular mRNA COVID-19 vaccine
- Aspirin: Study Finds Greater Benefits for These Colorectal Cancer Patients
- Cancer Can Occur Without Genetic Mutations?
- Statins Lower Blood Lipids: How Long is a Course?
- Warning: Smartwatch Blood Sugar Measurement Deemed Dangerous
- Mifepristone: A Safe and Effective Abortion Option Amidst Controversy
- Asbestos Detected in Buildings Damaged in Ukraine: Analyzed by Japanese Company
Mahidol University in Thailand develops chimeric Spike circular mRNA COVID-19 vaccine
- Red Yeast Rice Scare Grips Japan: Over 114 Hospitalized and 5 Deaths
- Long COVID Brain Fog: Blood-Brain Barrier Damage and Persistent Inflammation
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Mahidol University in Thailand develops chimeric Spike circular mRNA COVID-19 vaccine.
On March 28, 2022, the Patompon Wongtrakoongate group of Mahidol University in Thailand uploaded a preprinted article on BioRxiv: Broad neutralization of SARS-CoV-2 variants by circular mRNA producing VFLIP-X spike in mice.
They developed a broad-spectrum circular mRNA vaccine for COVID-19, designed with VFLIP Spike antigen and introducing 6 mutations from VOCs, which can trigger broad-spectrum serum neutralizing antibodies targeting various VOCs and TH1-biased cellular immunity reaction.
Compared with the widely used S-2P Spike antigen, the VFLIP Spike antigen has a glycosylation site closer to the natural Spike and has better stability.
On the basis of antigens based on structural rational design, the construction of chimeric mutant Spike, combined with the circular RNA platform, provides a very good idea for the development of iterative mRNA vaccines.
Antigen design
For antigen design, researchers used Erica Ollmann Saphire ‘s Spike antigen-VFLIP based on structure design . Compared with S-2P/S-6P, VFLIP not only has significantly improved expression and stability, but also has the same glycosylation site as native Spike. VFLIP antigen, modified mutations in the following three aspects on the Spike protein sequence:
(1) Five proline mutations ( V ) were introduced. Compared to SP-6, the lysine of the native Spike protein at position 986 is restored, thereby preserving the salt bridge mediated by K986.
(2) A short, flexible, linking peptide GGGSGGGS was introduced at the S1/S2 furin cleavage site to prevent S1 loss-triggered conformational transition after S2 fusion.
(3) Mutation Y707C and T883C introduced a disulfide bond between the S2 subunit of Spike monomer and the S2′ of another Spike monomer adjacent to it, so as to maintain the native trimer of Spike protein.
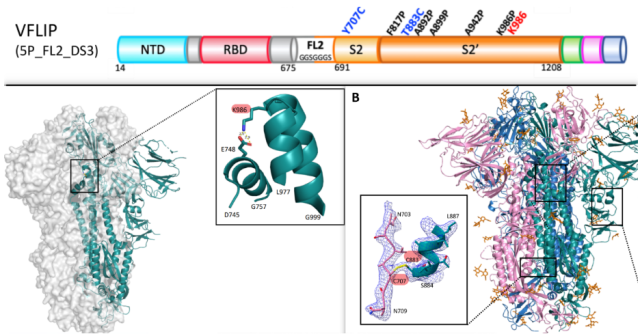
Erica Ollmann Saphire Structural Design-Based Spike Antigen—VFLIP.
Going back to this article, they used the VFLIP Spike antigen as the backbone, and on the basis of the original Spike sequence of D614G, they simultaneously introduced five mutations from the Beta and Delta RBD regions , namely K417N, L452R, T478K, E484K and N501Y , to form a mosaic . Synthesized mutant Spike antigen , called VFLIP-X .
At the same time, the researchers also constructed the H ex aPro-X Spike antigen , which consists of 6 proline mutations + 5 identical mutations from the Beta and Delta RBD regions, to compare the Spike caused by two different antigen designs. differences in protein expression.
The researchers drew the phylogenetic tree of the Spike sequences of various new coronavirus variants and the chimeric mutant Spike sequences, and found that the chimeric mutant Spike sequences were closer to the Omicron – B.1.1.529 Spike sequence on the branch .
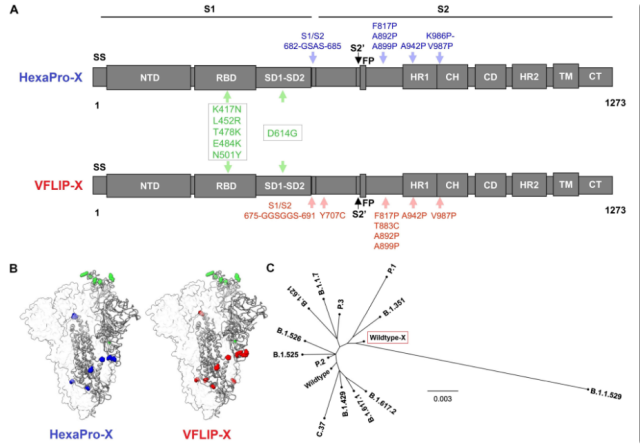
VFLIP Spike Antigen Design and HexaPro-X Spike Antigen Design and Spike Sequence Phylogenetic Tree Comparison.
CircRNA construction
After determining the antigen sequence, we used the Anabaena pre-tRNA type I intron-mediated circular RNA construction method developed by Daneil G. Aderson to construct the circular RNA stable and highly expressed protein , and construct CircRNA – VFLIP-X and CircRNA -HexaPro-X , transfected HEK293T cells, and found that the intracellular expression levels of circRNAs encoding two types of Spike proteins were similar.
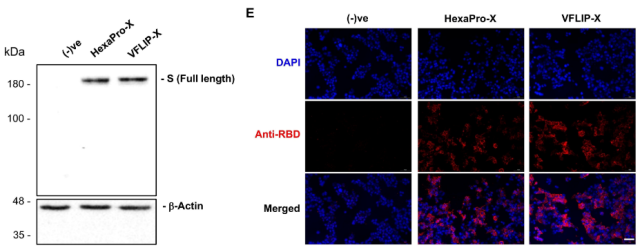
WB and immunofluorescence confirmed that the circRNAs encoding the two Spike antigens could be expressed normally in HEK293T cells.
Serum antibody response
Differences in serum antibody responses triggered by the two vaccines
CircRNA – VFLIP-X and CircRNA -HexaPro -X were encapsulated with LNP ( ionizable lipid SM102 ) , and mice were immunized with two doses of 5ug, 3 weeks apart. ELISA data showed that both vaccines triggered equal levels of serum-bound antibody IgG targeting Omircron-B.1.1.529 Spike.
Importantly, the pseudovirus neutralization assay showed that circRNA -VFLIP-X vaccine could trigger high titers of serum neutralizing antibodies targeting Omicron – B.1.1.529 , however , circRNA -HexaPro-X could not trigger targeting Serum neutralizing antibody to Omicron – B.1.1.529 .
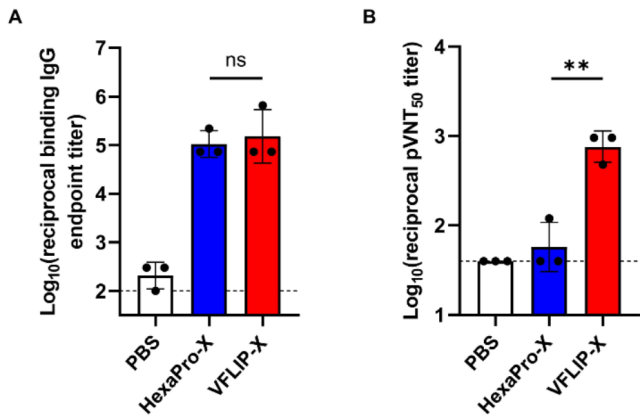
Two doses of CircRNA-VFLIP-X and CircRNA-HexaPro-X vaccines immunized mice (5ug), triggering similar levels of targeting Omicron-B.1.1.529 anti-Spike IgG serum-bound antibodies, while only CircRNA-VFLIP-X vaccine In order to trigger high titers of serum neutralizing antibodies targeting Omicron-B.1.1.529.
CircRNA -VFLIP-X triggers broad-spectrum neutralizing antibodies
The researchers immunized mice with CircRNA -VFLIP-X vaccine, divided them into a high-dose group of 5ug and a low-dose group of 1ug, collected serum, and tested the serum neutralizing antibody activity targeting various new coronavirus variants. It was found that 1ug in the low-dose group could not trigger serum neutralizing antibodies, but 5ug in the high-dose group could trigger high levels of broad-spectrum serum neutralizing antibodies targeting VOCs variants .
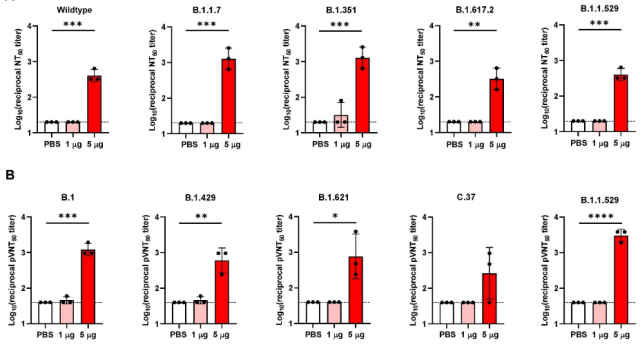
Both live virus and pseudovirus neutralization assays showed that the CircRNA-VFLIP-X vaccine triggered broad-spectrum seroneutralizing antibodies targeting various SARS-CoV-2 variants.
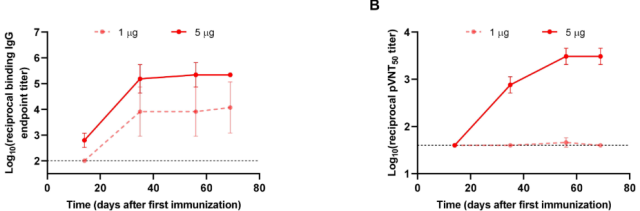
Two doses of CircRNA-VFLIP-X vaccine immunized mice with an interval of 3 weeks, and the titers of anti-Spike-IgG targeting B.1.1.529 and serum neutralizing antibody titers increased with time.
Cellular immunity
Seventh week after the second dose of CircRNA-VFLIP-X vaccine, spleen cells were isolated, and the spleen cells were stimulated with Omicron – B.1.1.529 – Spike polypeptide library, and the cellular immune response was analyzed by ELISpot. Spleen cells of vaccinated mice secreted high levels of IFN-γ , but very low levels of IL-4 .
ELSIA data showed that 5ug immunization of mice in the high-dose group triggered high levels of IgG2a and IgG1 .
The above data suggest that the CircRNA -VFLIP-X vaccine triggers a TH1-biased cellular immune response .
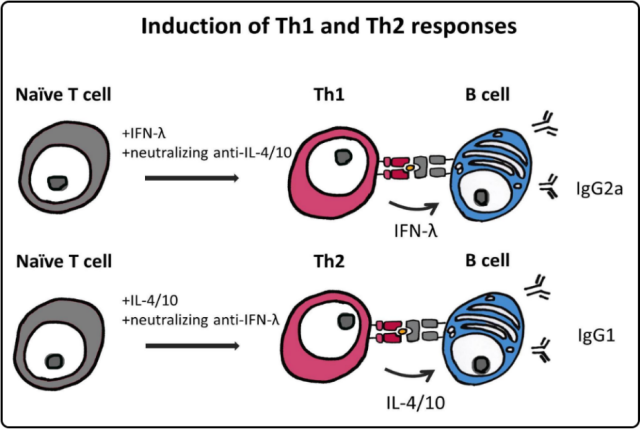
TH1 and TH2 cellular immune responses stimulate B cells to secrete different IgG subtypes, and the ratio of the two types of IgG antibodies characterizes the bias of T cell immune responses.
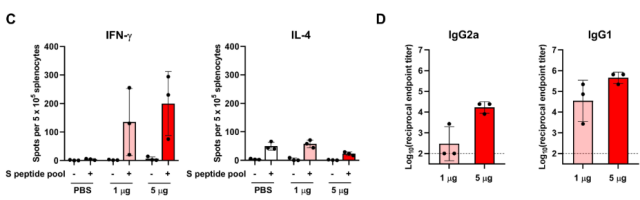
CircRNA-VFLIP-X vaccine triggers TH1-biased cellular immune responses
Summary
(1) The VFLIP-X Spike sequence designed in this study, based on the original Spike sequence of D614G, introduced five new coronavirus VOCs mutations in the RBD region, namely K417N, L452R, T478K, E484K and N501Y.
T478 and E484 are located in the peak region ( peak ) of the Spike-RBM motif, K417 and L452 are located in the valley region ( Valley ) of the Spike-RBM motif, and N501 is located in the flat-top region ( Mesa ) of the Spike-RBM motif .
The presence of these mutations in Alpha/Beta/Delta increases the binding affinity of Spike and ACE2, escaping the serum neutralization response triggered by the original strain.
The introduction of these five mutation sites enables the CircRNA -VFLIP-X vaccine to have a very broad-spectrum neutralizing ability in the face of VOCs.
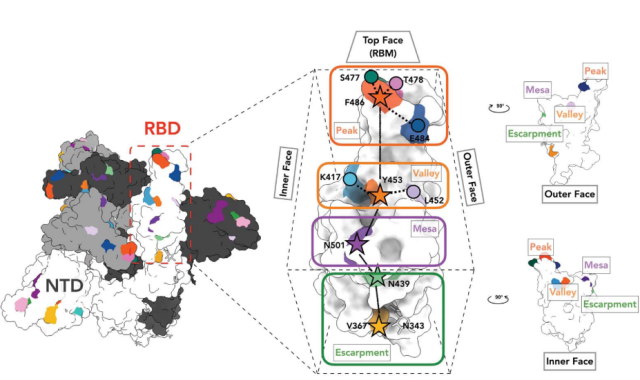
Important mutations in the RBD region in a trimer Spike with the RBD in the ascending conformation.
(2) Although CircRNA -HexaPro-X can be expressed normally in cells and trigger anti-Spike IgG serum-binding antibodies, it cannot trigger serum-neutralizing antibodies targeting Omicron-B.1.1.529.
In 2021, Danilo Casimiro published the article: Immunogenicity and efficacy of mRNA COVID-19 vaccine MRT5500 in preclinical animal models , and found that the mRNA vaccine encoding the Spike antigen of the original HexaPro strain could not trigger serum neutralizing antibodies in the rhesus monkey model .
However, the HexaPro Omicron Spike mRNA vaccine developed by the Wang Yucai team of the University of Science and Technology of China ( the S1/S2 furin cleavage site is not mutated ) and the HexaPro Omicron Spike antigen + GSAS furin cleavage site mutation mRNA vaccine used by Sidi Chen of Yale School of Medicine can trigger the target Antibodies were neutralized to the serum of Omicron-B.1.1.529 . Therefore, the effect of this HexaPro-Spike antigen design on the triggered serum neutralizing antibodies needs to be explored in more future articles.
(3) The prefusion conformation can trigger potent neutralizing antibodies, which has been confirmed. Notably, in the S-2P/Hexa antigen design, the RBD is more open .
However, other articles have shown that Spike antigens with the RBD fully closed , such as VFLIP, elicit stronger neutralizing antibody responses than S-2P/Hexa antigens .
Furthermore, VFLIP is more similar to native Spike in the distribution of glycosylation sites than Hexa antigen .
Therefore, in the design of Spike antigen, how to balance the open conformation and closed conformation of RBD, and the relationship between the stable state and unstable state before fusion is a difficult problem that needs to be overcome.
Mahidol University in Thailand develops chimeric Spike circular mRNA COVID-19 vaccine
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



