British MHRA approves Amgen revolutionary lung cancer drug Sotorasib
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
British MHRA approves Amgen revolutionary lung cancer drug Sotorasib, targeting KRAS.
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
British MHRA approves Amgen revolutionary lung cancer drug Sotorasib, targeting KRAS.
Introduction:
From the discovery of a series of compounds that specifically bind to the KRAS G12C mutant in a paper that shocked the medical community in 2013, to the approval of the new drug Sotorasib by the FDA, it took eight years for Amgen to complete. Based on the FDA’s “Orbis Plan”, which realizes simultaneous approval of cancer drug applications in multiple countries, the British HSA recently followed up and approved Amgen, a revolutionary lung cancer drug. Earlier, Amgen announced that Sotorasib has been certified as a “breakthrough therapy drug” in China, and more patients will benefit from this new type of treatment in the future.
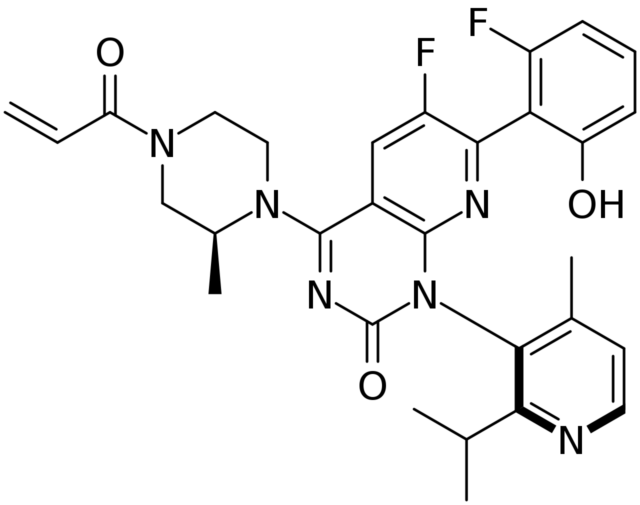
Chemical structure of Sotorasib
Lung cancer is currently one of the leading causes of cancer deaths worldwide. Among the major lung cancer driver gene mutations, the “KRAS G12C” mutation in KRAS occurs in approximately 14% of non-small cell lung cancer (NSCLC) patients. For 30 years, the target KRAS, which has been called “non-drugable” by the pharmaceutical industry, has a complex structure and is highly similar to other members of the RAS family, HRAS and NRAS.
All this changed in 2019, when Amgen announced the results of its groundbreaking Phase 1 trial of its KRAS G12C inhibitor, which showed good prospects in the Phase 1 trial, indicating a disease control rate of 55.2%. In May 2021, the FDA approved Amgen’s Lumakras (Sotorasib) for the treatment of locally advanced or metastatic non-small cell lung cancer (NSCLC) patients with KRAS G12C mutations, thus making history.
This approval is based on the positive results of the cohort of patients with advanced NSCLC in the Phase II CodeBreaK 100 study. In the phase II trial, 124 lung cancer patients with KRAS G12C mutations took 960 mg of Sotorasib orally every day. The results showed that the disease control rate (DCR) reached 81%, and the median duration of response (DoR) was 10 months. Importantly, 36% of patients had a significant reduction in tumor size (≥30%).
On September 10, the British Medicines and Healthcare Products Administration (MHRA) approved Lumakras (sotorasib) for the treatment of patients with advanced NSCLC with KRAS G12C mutations. The approval is based on shared data from the Orbis project, an initiative of the FDA’s Oncology Center of Excellence. The Orbis project is an international partnership between global drug regulatory agencies, dedicated to accelerating the acquisition of innovative cancer drugs.
“Sotorasib drug is one of the most exciting breakthroughs in the field of lung cancer treatment in 20 years. It targets a cancer gene that could not be targeted before. It is based on decades of laboratory research that reveals cancer. “The internal workings of the United Kingdom,” said Professor Charles Swanton, Chief Clinician of Cancer Research UK, “This drug expands our list of effective and precise treatments for lung cancer, which can help improve survival for patients with limited options. Good news Yes, patients in the UK will now benefit from this new type of treatment.”
After obtaining MHRA approval, the National Health Service (NHS) and Amgen reached an agreement to enable eligible lung cancer patients in the UK to obtain this revolutionary drug as soon as possible. Because it is an early use agreement, it is expected that approximately 600 patients in the UK will benefit from it each year. At the same time, the National Institute of Health and Care Excellence (NICE) will continue to conduct ongoing evaluations of the drug. The final NICE guidelines are expected to be released in March 2022.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



