Climate change: Alpine soil microbial functions and biogeochemical cycles
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Climate change: Alpine soil microbial functions and biogeochemical cycles
Climate change: Alpine soil microbial functions and biogeochemical cycles . Climate change alters temporal dynamics of alpine soilmicrobial functioning and biogeochemical cycling via earlier snowmelt.
Based on the results of this study, we found that mutations in soil microbial community composition during snowmelt are closely related to changes in a series of microbial functions and biogeochemical cycles, and this seasonal mutation is mainly driven by earlier snow melting.
Guide
Soil microbial communities play a regulatory role in the global biogeochemical cycle and respond quickly to changing environmental conditions. However, understanding how soil microbial communities respond to climate change and how this affects biogeochemical cycles remains a major challenge. The speed of climate change in high mountain areas is twice the global average, which will greatly reduce the snow cover, and the snow cover will melt in advance in spring. This feature makes relevant research particularly important in this region.
In this study, we found that the spring snowmelt triggered a mutation in the composition of the soil microbial community in the alpine grassland. This mutation is closely related to changes in soil microbial functions, biogeochemical pools and fluxes. In addition, through experimental control of snow cover, we have shown that snow melting drives this sudden seasonal shift in a wide range of microbial and biogeochemical soil properties.
The previous winter conditions did not change the process that occurred during the snowmelt. Our findings emphasize the importance of seasonal dynamics to soil microbial communities and their regulation of biogeochemical cycles. In addition, our findings indicate that the melting of early spring snow caused by climate change will have a profound impact on the microbial communities and nutrient cycles in these globally distributed alpine ecosystems.
Paper ID
- Original name: Climate change alters temporal dynamics of alpine soilmicrobial functioning and biogeochemical cycling via earlier snowmelt
- Journal: The ISME Journal
- IF: 9.180
- Posting time: 2021.02.22
- Corresponding author: Arthur A. D. Broadbent
- Corresponding author unit: The University of Manchester (The University of Manchester)
Experimental design
The experimental site for this study was selected in an alpine grassland near Obergurgl. At this site, this study selected 15 5 m×5 m plots and randomly assigned three treatments (5 plots for each treatment): snow removal treatment, snow addition treatment, and untreated blank plots. A total of 4 snow control treatments were carried out throughout the test cycle. Each time, a snow blower or a shovel was used to remove the snow removed from the snow block to a block less than 10 cm deep, and then this part of snow was evenly added to the added snow. In the processed plots, the unprocessed blank plots did not cause any interference. During the test, we recorded the exact melting date of each plot, that is, the snow melting time. Through records, we found that the snow-cover regulation treatment changed the snowmelt time and had a significant impact on soil temperature and moisture.
We sampled at 6 time nodes that span the seasonal transition from the end of winter to the beginning of summer in 2017, namely the end of winter (March 28; “Winter 28/3”) and the snowmelt period (June 1 and 8, “Snowmelt 1/6” and “Snowmelt 8/6”), spring (June 12th and 18th, “Spring 12/8” and “Spring 18/6” respectively), and early summer (July 8 Day; “Summer 8/7”). In order to avoid edge effects, each sampling is selected at least 1 m from the edge of the plot. Whenever possible, use a steel sampler to collect soil cores with a diameter of 2 cm and a depth of 7 cm from five randomly selected locations on each sample plot (some soil cores have a shallower sampling depth when the soil is frozen). ), take out the vegetation and litter after each sampling, and then mix them evenly. Five sub-samples of approximately 200 mg were collected from each plot, lysed on site, and then stored at -80°C for molecular experiments. The remaining soil samples were passed through a 4 mm sieve for enzyme and physiological and biochemical analysis.
Results:
1 Seasonal dynamics of soil microbial communities and biogeochemical cycles
Whether it is the abundance and composition of soil bacterial and fungal communities, or the relative abundance and potential enzyme activity of enzyme genes that catalyze the key processes of the C and N cycles, there is a clear transition from winter to summer. In addition, the composition and function of soil microbial communities change with changes in biogeochemical pools and fluxes.
Based on phospholipid fatty acid (PLFA) technology to label soil fungi and bacteria, we found that the active microbial biomass in the soil decreased by 88% from late winter to summer, and fungi and bacteria showed similar declines (Figure 1a-b) ). During the entire sampling period, bacteria are more abundant than fungi.
This phenomenon is especially obvious during the snowmelt period. At the same time, we also found that gram-negative bacteria have more advantages than gram-positive bacteria. The composition of the soil microbial community assessed by molecular barcoding method showed that there are significant differences between the seasons of bacteria and fungi at the mesh level (Figure 1c-d). It is worth noting that the relative abundance of Acidobacteriales increased by 301% from winter to summer, and its relative abundance ranked second.
In contrast, Actinomycetales (the second most abundant order of bacteria during winter) has a 42% decrease in relative abundance from winter to summer. In the fungus kingdom, from winter to summer, the relative abundance of Pleosporales, Atheliales, and Pezizales decreased by 50%, 71%, and 88%, respectively, while the relative abundance of Geminibasidiales increased by more than 8000%. From late winter to summer, the relative abundance of Helotiales, which is mainly related to roots, increased by 93% and became the dominant fungus.
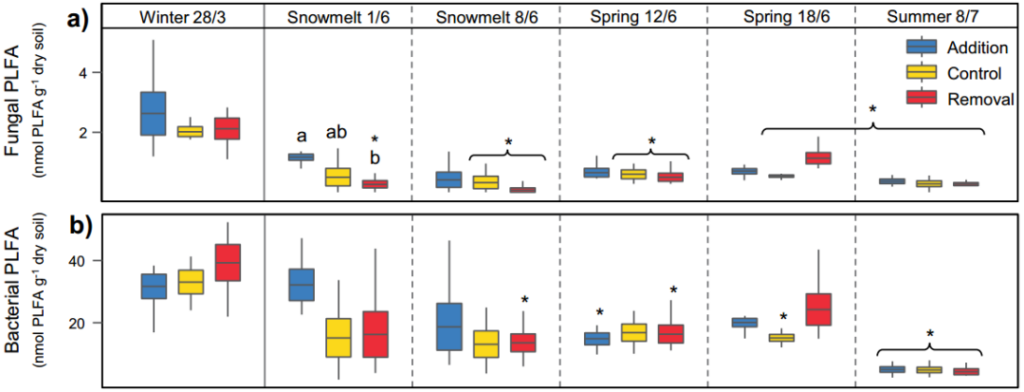

Figure 1 The influence of seasonal changes and snow cover treatments on the composition of soil microbial communities. Mark soil fungi (a) and bacteria (b) abundance based on PLFA technology, where only the microbial groups with significant differences (p<0.05) are shown in the figure; cd stands for bacteria (c) and fungi (d) in the table The average relative abundance at the level.
Seasonal changes in soil microbial community composition are accompanied by significant changes in potential microbial functions. The relative abundance of various enzyme-encoding genes involved in the key pathways of the N cycle increased significantly in summer (Figure 2a). In addition, nitrification genes including ammonia oxidizing archaea (AOA) genes amo A, amo B, and amo C, as well as nitrite oxidation genes including nxr A and nxr B, all increased significantly (Figure 2a) . The relative abundance of nitrogen fixation genes (nif K), denitrification genes (nir K and nos Z), nitrate reduction genes (NR, nar H, and nar I) and organic nitrogen synthesis genes (gdh_K00261 and gdh_K00262) are also small but Significant increase. Similarly, the relative abundance of the 9 subfamilies of cellobiohydrolase (CBH), β-glycosidase (GLC) and β-xylosidase (XYL), which play an important role in the C cycle, increased in summer, while in 7 GH43 Among the subgroups, only the relative abundance of XY1 decreases in summer.
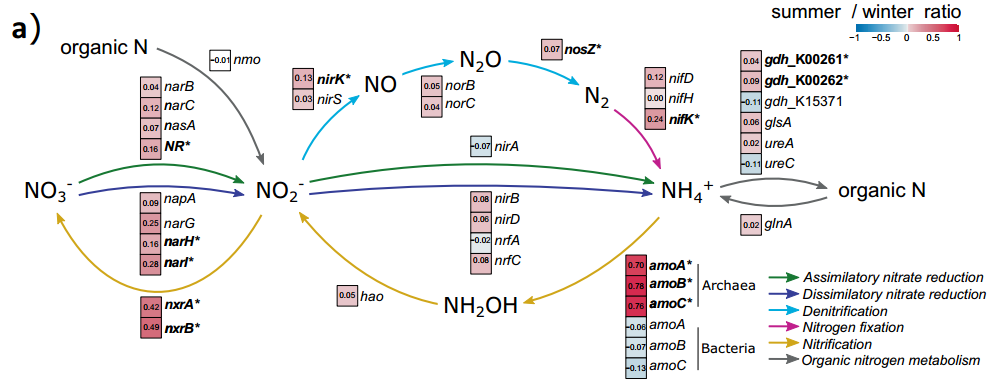
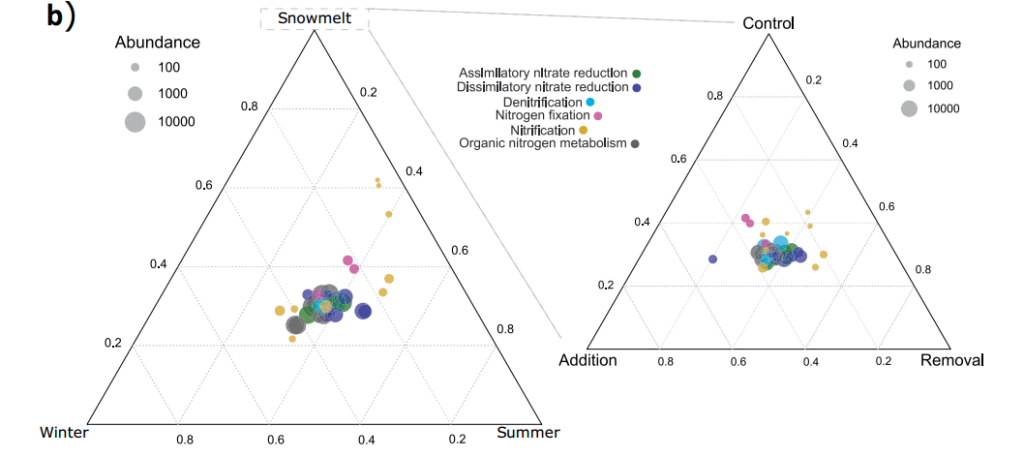
Figure 2 The complete nitrogen cycle pathway reconstructed based on the relative abundance of genes. Each pathway in a represents the log ratio between summer and winter. The white box represents no change, the red box represents the higher potential in summer, and the blue box represents the higher potential in winter. Activity; b represents the distribution and abundance of nitrogen cycle genes in different seasons (left) and the distribution and abundance of nitrogen cycle genes under snowmelt treatment (right). Different pathways are depicted in different colors, and the size of the circle indicates the relative abundance of genes Degree size.
Most of the enzyme activities and phosphatase (PHO) activities related to the C cycle increased by 118-274% in summer. However, compared with winter, their potential activity decreases in summer. Potential urease (URE) and N-acetylglucosaminidase (NAG) activities also had similar declines (Figure 3a). On the contrary, the potential activities of phenol oxidase (POX) and peroxidase (PER) detected in summer increased by more than 50% compared with winter (Figure 3b), while their microbial biomass-specific activities increased by more than 1000% . Soil respiration is a comprehensive index to measure soil activity. It reaches the highest directly after the snow melts, but decreases in summer.
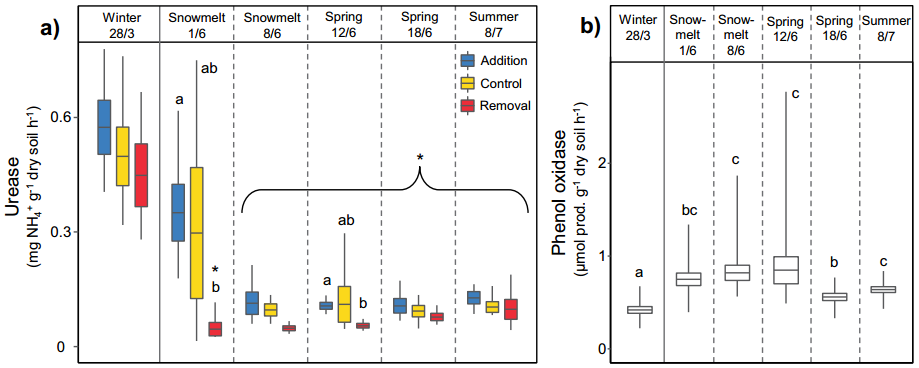
Figure 3 Seasonal change and snow cover
Management of the soil
The effect of extracellular enzyme activity. a-b represent the potential enzymatic activity of urease (a) and phenol oxidase (b), respectively.
Along with the observed changes in the composition and function of soil microbial communities, soil properties have also undergone significant changes. From the end of winter to summer, soil pH (from 6.8 to 4.9), available NH4+ (reduced by 99%), soluble organic nitrogen (DON, a decrease of 83%), and total soluble organic carbon (DOC, a decrease of 92%) ) And soil water content (a decrease of 44%) have a significant decline (Figure 4a-c). On the contrary, compared with the end of winter, the ratio of available NO3- and DON to available inorganic nitrogen in summer increased by 46% and 329%, respectively (Figure 4d). In addition, during the 14-day culture under the experimental conditions of 25°C, the rate of ammoniation and nitrification varied from -22±4 and 60±7 µg N g-1 dry weight/day in winter to 1.7±0.2 and nitrification rate in summer, respectively. -0.4±0.1 µg N g-1 dry weight/day.
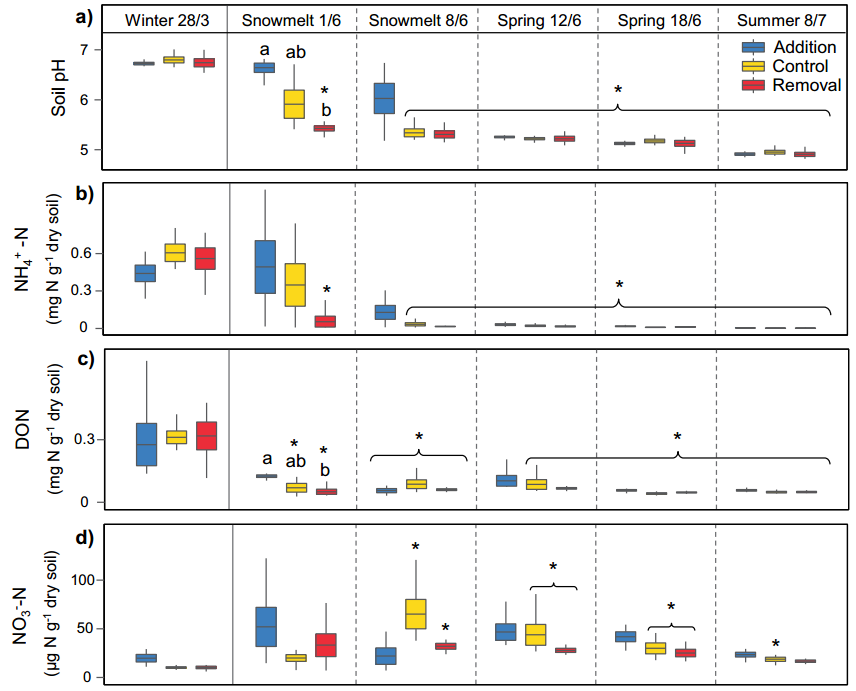
Figure 4 The effect of seasonal change and snow treatment on soil properties and pH. a-d represent soil pH (a), ammonium nitrogen concentration (b), soluble organic nitrogen (c) and nitrate nitrogen concentration (d), respectively.
2 The influence of snowmelt time and previous winter conditions
The treatment of snow cover significantly affected the composition and function of soil microbial community and the time of biogeochemical cycle. Compared with the control and snow addition treatments, the snow removal treatment advanced the snowmelt time by 3 and 10 days on average. Compared with the control, snow removal led to an early reduction in microbial biomass and an early change in community composition, which was related to the potential activities of URE, NAG, GLC, CBH and XYL (Figure 3a-b), soil pH and available NH4+ (Figure 4a-b). ) Is related to the advance reduction. Compared with the snow addition treatment, the snow removal treatment continuously reduced the specific urease activity of the microbial biomass and continuously increased the ratio of DON to available inorganic nitrogen. Although we have detected some genes that are responsive to snow cover regulation, the relative abundance of these genes is not strongly affected by snow cover regulation. Genes involved in the carbon and nitrogen cycles show significantly higher relative abundance in summer (for example, amo A, amo B, and amo C; Figure 2a), and this phenomenon also occurs during snow removal during snowmelt (Figure 2b) ), and genes that show higher relative abundance in winter tend to show higher abundance during the snow-melting period (1/6).
During the second sampling period (1/6 of the snowmelt), the snowmelt time was significantly (p<0.05) explaining the 40-85% variation of the 22 measured soil parameters (this sampling time corresponds to the snow melting of most of the plots) , Which is precisely the time point where the difference between treatments is greatest). These indicators include: total biomass, fungal biomass and bacterial biomass; the abundance of fungi relative to bacteria; the relative abundance of bacterial orders Ellin6513 and Ellin5290 and fungal orders Pezizales and Geminibasidies; the potential activities of six enzymes (URE, NAG) , PHO, CBH, XYL and GLC); the microbial biomass-specific activities of POX and PER; and a series of soil non-biological properties including pH, DOC, and effective NH4+. At this point in time (1/6 of the amount of snow melt), 80% of the plots with snow removal treatment have completely melted, while the plots with snow treatment have not yet melted.
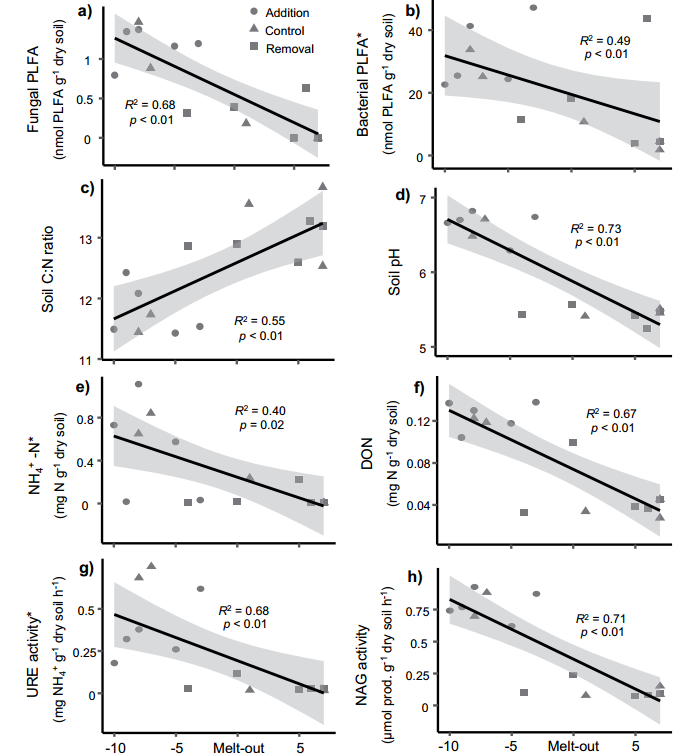
Figure 5 The influence of snowmelt time on the composition, function and biogeochemical cycle of soil microbial communities during the seasonal transition period. ah represents the abundance of fungi (a) and bacteria (b) based on PLFA markers, soil carbon to nitrogen ratio (c), soil pH (d), soil ammonium nitrogen concentration (e), soil soluble organic nitrogen content (f) , Soil urease activity (g) and NAG enzyme activity (h).
The previous winter conditions had no significant effect on soil microorganisms or biogeochemical processes, and had nothing to do with the snowmelt time. Among all the parameters we measured, there was no significant difference between the snow removal treatment of 01/06, the control treatment of 08/06 and the snow addition treatment of 12/06 (Figure 1-4, p<0.05).
Discussion:
Our data show that the spring snowmelt in the alpine steppe triggered a sudden change in the composition of the soil microbial community, and was accompanied by a series of simultaneous changes in soil microbial functions, biogeochemical pools and fluxes. Our findings also indicate that climate change in high mountains will lead to a reduction in snow cover, which in turn promotes this seasonal shift. This study found that the relatively moderate advancement of the snowmelt time is of great ecological significance, but it is expected that there will be a more extreme advancement of the snowmelt time at the end of this century. Our findings indicate that these advances in the time of snowmelt in the future will have a profound impact on the C and N fluxes, the composition of plant communities, and the productivity of ecosystems in alpine grasslands that are widely distributed around the world.
This book explores the temporal changes of soil microbial community composition under field conditions, and clearly associates it with changes in soil microbial functions and biogeochemical cycles, thereby promoting the understanding of microbial ecology and biogeochemistry. The higher microbial biomass in winter is related to the higher soil enzyme activity potential. This shows that, like other ecosystems, the key microbial-mediated biogeochemical processes in the alpine grasslands have been continuing even under snow cover.
The results of this study show that the soil microbial community in winter helps to decompose organic compounds including xylan, cellulose and chitin. In addition, the dominant microbial groups in the soil microbial community in winter are closely related to their functions. Specifically, the order of fungi Thelebolales, the order of bacteria Actinomycetales and Bacilliales are all closely related to cellulose degradation. Cellulose fibers are difficult to directly contact, but the mycelial growth of fungi and cellulolytic bacteria can penetrate the pores of plant cell walls, so that cellulase and cellulose polymers are in close contact. The potential enzyme activity is higher in winter, and the specific enzyme activity of microbial biomass is higher in summer.
This suggests that high microbial biomass may explain the potential high enzyme activity in winter, but microorganisms are usually used more for enzyme production in summer, especially It is the lignin degrading enzymes POX and PER. The pH value (5.0) of the buffer solution used in this study is very close to the in-situ pH value of summer soil (average = 4.9), but lower than the pH value of winter soil (average = 6.8). Considering the influence of soil pH on enzyme activity, we may have some deviations in the snow-adding treatments during winter and snowmelt compared with the results in summer (Figure 4a). Nevertheless, the stable soil temperature and moisture under the snow cover, high substrate availability, and high microbial biomass indicate that although the in situ enzyme activity rate of soil enzymes may be lower than the rate measured in the laboratory, the total winter Enzyme activity still maintains a high level.
The composition of the soil microbial community in summer is closely related to the key functions that occur in summer. Specifically, Acidobacteriales and Solibacterales, which are closely related to acidic soil and lignin degradation, dominate the soil. This explains exactly why lignin-degrading enzymes POX and PER have higher potential activities in summer. The degradation of lignin may reflect that due to severe nitrogen limitation, microorganisms will use nitrogen from the more recalcitrant organic matter pool, which corresponds to the sharp drop in DON (−83%) and effective NH4+ (−99%). We also detected relatively high abundance of genes involved in nitrification, especially amo A, amo B and amo C of AOA. The increasing importance of AOA may reflect the low soil pH in summer and the high nitrification remaining after snowmelt driven by large amounts of NH4+ accumulated in winter. Considering that the nitrification process can produce H+ ions, the high nitrification rate during the snow melting process may be one of the reasons that cause the soil pH to drop significantly. Due to the decrease in bacterial and fungal abundance, DON and NH4+ during the snowmelt period, we have observed a significant decrease in microbial biomass and soil nitrogen availability in summer. In addition to the decline in microbial biomass and soil N availability, snowmelt also triggers changes in microbial composition, potential enzyme activities involved in the C and N cycles (such as URE, NAG, CBH, XYL, and GLC), and soil pH. The results show that the spring snowmelt plays a central role in controlling the temporal dynamics of soil microbial community composition, and the temporal dynamics of soil microbial community composition are closely related to changes in soil nitrogen cycling under field conditions in alpine grasslands.
Our findings indicate that changes in the time of snowmelt in spring have changed the time of seasonal changes in the composition, function, and biogeochemical cycle of soil microbial communities in the alpine steppe. However, in this study, we did not find that the previous winter conditions have an impact on the microbial and biogeochemical processes that occur during snowmelt. On the contrary, the main mechanism for the reduction of snow cover in winter has an impact on the ecology because the earlier snowmelt leads to the earlier transformation of the soil microbial community and biogeochemical cycle. As the snow cover decreases, seasonal changes in soil microbial community composition, functions, and biogeochemical cycles may occur earlier, which improves our understanding of soil microbial response to climate change. Winter soil microbial communities in alpine regions play important ecosystem functions, such as decomposing complex litter residues, and the related accumulation of high soil DON and effective NH4+. However, under future climate change conditions, these functions are likely to be affected. reduce. This may lead to a potential mismatch between seasonal soil N flux and the beginning of plant growth in spring, thereby affecting the C and N fluxes of the ecosystem.
Sum up
Based on the results of this study, we found that mutations in soil microbial community composition during snowmelt are closely related to changes in a series of microbial functions and biogeochemical cycles, and this seasonal mutation is mainly driven by earlier snow melting. Taking into account that climate change is expected to lead to reduced snow cover and earlier snow melting, our results indicate that the transition of soil microbial communities from winter to summer will occur earlier this year. This may lead to changes in the annual carbon and nitrogen fluxes of seasonal snow ecosystems. Although the snowmelt time obtained by our experimental control has significant ecological differences, our data is relatively moderate compared to the predicted value at the end of this century (the maximum number of days in advance is 10 days), and according to the prediction of snowmelt in the European Alps It is expected to be 50-130 days in advance. Changes in the time of snowmelt on this scale may have a profound impact on the biogeochemical cycles and plant growth in these fragile global ecosystems.
(source:internet, reference only)
Disclaimer of medicaltrend.org



