Johns Hopkins found Clostridium difficile promoted bowel cancer for the first time
- Normal Liver Cells Found to Promote Cancer Metastasis to the Liver
- Nearly 80% Complete Remission: Breakthrough in ADC Anti-Tumor Treatment
- Vaccination Against Common Diseases May Prevent Dementia!
- New Alzheimer’s Disease (AD) Diagnosis and Staging Criteria
- Breakthrough in Alzheimer’s Disease: New Nasal Spray Halts Cognitive Decline by Targeting Toxic Protein
- Can the Tap Water at the Paris Olympics be Drunk Directly?
Johns Hopkins found Clostridium difficile promoted bowel cancer for the first time
- Should China be held legally responsible for the US’s $18 trillion COVID losses?
- CT Radiation Exposure Linked to Blood Cancer in Children and Adolescents
- FDA has mandated a top-level black box warning for all marketed CAR-T therapies
- Can people with high blood pressure eat peanuts?
- What is the difference between dopamine and dobutamine?
- How long can the patient live after heart stent surgery?
Johns Hopkins found Clostridium difficile promoted bowel cancer for the first time.
Colorectal cancer causes 1.9 million new patients every year, seriously endangering human health, and it is the third largest cancer in the world [1]. Risk factors include physical inactivity, Western-style diet, smoking, and obesity.
In recent years, it has been found that the incidence of colorectal cancer is also closely related to the intestinal flora.
Enterotoxigenic Bacteroides fragilis[2], pks+Escherichia coli[3] and Fusobacterium nucleatum[4] in the gut have been identified one after another to promote the occurrence and development of colorectal cancer.
Recently, a new member has been added to the blacklist of cancer-promoting intestinal bacteria.
A team led by Cynthia L. Sears of Johns Hopkins University discovered for the first time that a toxigenic strain of Clostridium difficile in the mucosal sera of patients with colorectal cancer can induce colon tumors in mice, and its cancer-promoting effect depends on the continuous colonization and production of the strain The toxin TcdB. Related papers were published in Cancer Discovery , a top journal in oncology [5].
In previous studies, the Cynthia L. Sears team found that the human gut microbiota, whether from colorectal cancer patients or healthy people, formed biofilms on the surface of the intestinal mucosa, which could induce colon tumors in mice [6] .
In this study, the researchers continued their experiments to analyze the cancer-promoting components and detailed mechanisms of the gut microbiota.
First, the researchers obtained bacterial-rich mucosal serum from patients with colorectal cancer and inoculated it into mice by gavage.
They found that the number of colon tumors in mice with colorectal cancer increased significantly, verifying the strong promotion of intestinal flora on colorectal cancer. cancer effect.
By microbial sequencing of the intestinal flora of mice, the researchers found that compared with healthy mice, mice with colorectal cancer had some unique intestinal bacteria, and these intestinal bacteria were transplanted into mice without cancer. mice, can strongly induce colorectal cancer in mice.
However, these unique gut bacteria do not include known cancer-promoting enterobacteria. This suggests that other new intestinal bacteria are playing a role in promoting cancer!
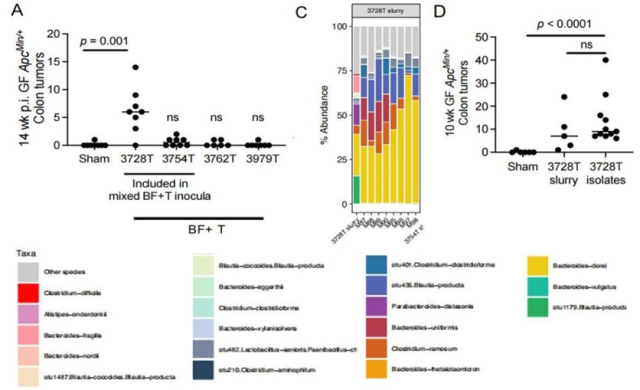
Panel A: The number of colon tumors in a colorectal cancer mouse model inoculated with intestinal mucosal serous from a colorectal cancer patient (3728T) was significantly increased; Panel C: Changes in intestinal flora in colorectal cancer mice; Panel D: Transplanted special intestines The number of colon tumors was also significantly increased in mice with tract microbiota
The researchers tested these intestinal bacteria and found that C. difficile was significantly enriched in colon tissue and feces of tumor-inducing mice, and the relative abundance in feces was positively correlated with the number of colon tumors.
In addition, RNA-sequencing revealed that the expression of C. difficile toxins TcdA and TcdB were also significantly upregulated in the colons of tumor-induced mice compared to their expression in healthy mice.
Clostridium difficile is an anaerobic bacterium that secretes TcdA (enterotoxins) and TcdB (cytotoxins) and causes disease through toxins.
From this, the researchers speculate that C. difficile may be the driver of tumorigenesis in mice with colorectal cancer.
To test whether C. difficile can drive colorectal carcinogenesis, the researchers removed C. difficile from the mucosal serous of colorectal cancer and then inoculated it into mice .
Significantly promote colon tumorigenesis. These results suggest that C. difficile is an essential factor driving tumorigenesis.
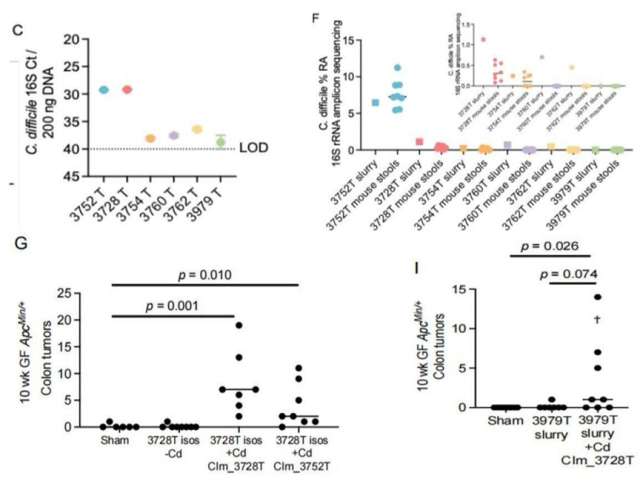
Clostridium difficile (C. difficile) is higher in intestinal mucosal mucus (Panel C) and tumorigenic mouse feces (Panel F) of colorectal cancer patients (3752T and 3728T); Figure G: tumor-promoting mucus removed C. difficile post-treated mice could not induce tumorigenesis; Figure I: non-tumor-promoting mucosal mucus post-treated mice with C. difficile could significantly induce tumorigenesis
Next, the researchers explored the mechanism by which Clostridium difficile promotes colon cancer.
The early response of the intestinal mucosa to cancer-promoting bacteria is a key step in the process of colon tumorigenesis [7]. To identify early tumorigenic events caused by C. difficile, the researchers examined the histological response of the colonic mucosa of mice after inoculation with C. difficile and found that abnormal crypts and microadenomas in the colon of mice gradually increased with the time of inoculation.
When increased, enterobacterial biofilms formed on the surface of the colon and invaded into the crypts.
Considering the significant cancer-promoting effect of C. difficile, the researchers expected C. difficile to be the main component of the gut microbiota of tumorigenic mice. The relative abundance of C. difficile is very low, and it is not the dominant species.
The researchers then used antibiotics to eliminate C. difficile, which almost completely blocked the formation of microadenomas after 2 weeks.
These results suggest that it is not the dominance of C. difficile in the microbiota, but rather sustained colonization and toxin production, that is key to promoting tumorigenesis.
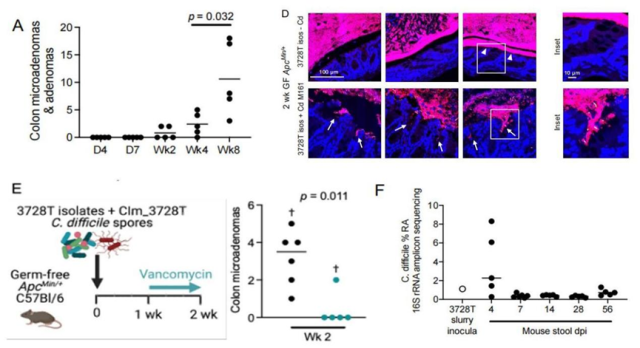
In mice inoculated with C. difficile, intestinal microadenomas gradually increased (Panel A), and enterobacterial biofilms formed on the surface of the colon and invaded into crypts (Panel D), but C. difficile was not a tumor-forming mouse intestine The dominant species of the tract flora (Panel F), elimination of C. difficile with antibiotics almost completely blocked the formation of microadenomas (Panel E).
TcdB is the most critical toxin in C. difficile infection, and C. difficile alone is sufficient to cause enteritis [8]. Therefore, the researchers examined the role of TcdB in C. difficile promoting bowel cancer.
Knock out TcdB of C. difficile, construct TcdB- strain, and inoculate mice with TcdB+ strain respectively, the researchers found that the number of colon tumors in mice with TcdB- strain was significantly reduced, and the formation of adverse lesions was significantly weakened.
In addition, knocking out TcdB did not Affects biofilm formation and Clostridium difficile colonization levels. These results demonstrate that TcdB is necessary for C. difficile to induce tumorigenesis.
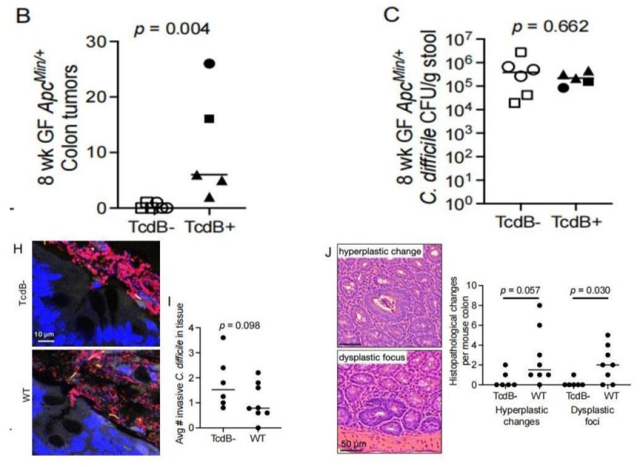
Knockdown of TcdB significantly reduced the tumor-promoting effect of C. difficile on colon tumors (Panels B and J), but did not affect biofilm formation (Panel H) and the level of C. difficile colonization (Panel C).
Finally, the researchers explored the cancer-promoting signaling pathway of C. difficile.
Single-cell sequencing showed that Wnt signaling in colonic progenitor cells was significantly activated in mice with colon cancer caused by C. difficile, and the production of reactive oxygen species in differentiated colon cells was significantly increased.
Wnt signaling is a hallmark-driven pathway of colorectal cancer, and oxidative stress is also closely related to the pathogenesis of colorectal cancer.
Gene enrichment analysis showed that the expression of oncogenes such as Myc was up-regulated, and the expression of tumor suppressors such as P53 was down-regulated.
Immune infiltration analysis showed a marked increase in the abundance and activation of lymphocytes characterized by IL-17 production.
These results suggest that C. difficile-induced colorectal carcinogenesis is associated with alterations in colonic epithelial cell transcriptional responses and oncogenic mucosal immunity.
Collectively, using human colorectal cancer specimens and mouse models, the researchers found that the intestinal pathogen C. difficile promotes colorectal cancer development, and its tumor-promoting effect is dependent on the C. difficile toxin TcdB, which is associated with the induction of Wnt signaling. , reactive oxygen species and cancer-promoting mucosal immune responses, revealing for the first time that chronic infection with Clostridium difficile is also a potential driver of colon tumor pathogenesis.
There are a large number of microorganisms in the human gut, which are closely related to the health of the human body and affect all aspects of the gut.
With the deepening of people’s understanding of the intestinal flora, the blacklist of cancer-promoting enterobacteria is also expanding, and Clostridium difficile may not be the last one.
references
[1] Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71(3):209-249. doi: 10.3322/caac.21660
[2] Tilg H, Adolph TE, Gerner RR, Moschen AR. The Intestinal Microbiota in Colorectal Cancer. Cancer Cell. 2018;33(6):954-964. doi:10.1016/j.ccell.2018.03.004
[3] Pleguezuelos-Manzano C, Puschhof J, Rosendahl Huber A, et al. Mutational signature in colorectal cancer caused by genotoxic pks+ E. coli. Nature. 2020;580(7802):269-273. doi:10.1038/s41586- 020-2080-8
[4] Gao Y, Bi D, Xie R, et al. Fusobacterium nucleatum enhances the efficacy of PD-L1 blockade in colorectal cancer [published correction appears in Signal Transduct Target Ther. 2021 Dec 21;6(1):434]. Signal Transduct Target Ther. 2021;6(1):398. Published 2021 Nov 19. doi:10.1038/s41392-021-00795-x
[5] Drewes JL, Chen J, Markham NO, et al. Human colon cancer-derived Clostridioides difficile strains drive colonic tumorigenesis in mice [published online ahead of print, 2022 Jun 9]. Cancer Discov. 2022;candisc.1273.2021-10 -4 15:58:09.930.doi:10.1158/2159-8290.CD-21-1273
[6] Tomkovich S, Dejea CM, Winglee K, et al. Human colon mucosal biofilms from healthy or colon cancer hosts are carcinogenic. J Clin Invest. 2019;129(4):1699-1712. Published 2019 Mar 11. doi: 10.1172/JCI124196
[7] Thiele Orberg E, Fan H, Tam AJ, et al. The myeloid immune signature of enterotoxigenic Bacteroides fragilis-induced murine colon tumorigenesis. Mucosal Immunol. 2017;10(2):421-433. doi:10.1038/mi. 2016.53
[8] Carter GP, Chakravorty A, Pham Nguyen TA, et al. Defining the Roles of TcdA and TcdB in Localized Gastrointestinal Disease, Systemic Organ Damage, and the Host Response during Clostridium difficile Infections. mBio. 2015;6(3): e00551. Published 2015 Jun 2. doi:10.1128/mBio.00551-15
Johns Hopkins found Clostridium difficile promoted bowel cancer for the first time.
(source:internet, reference only)
Disclaimer of medicaltrend.org
Important Note: The information provided is for informational purposes only and should not be considered as medical advice.



